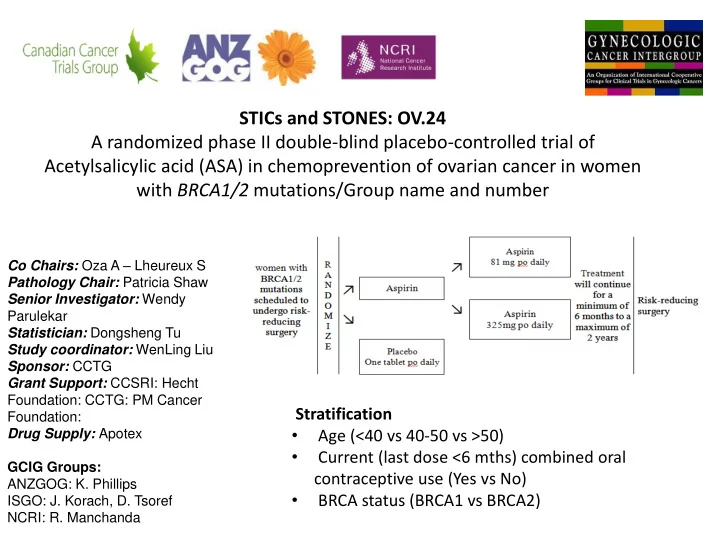
STICs and STONES: OV.24 A randomized phase II double-blind placebo-controlled trial of Acetylsalicylic acid (ASA) in chemoprevention of ovarian cancer in women with BRCA1/2 mutations/Group name and number
Co Chairs: Oza A – Lheureux S Pathology Chair: Patricia Shaw Senior Investigator: Wendy Parulekar Statistician: Dongsheng Tu Study coordinator: WenLing Liu Sponsor: CCTG Grant Support: CCSRI: Hecht Foundation: CCTG: PM Cancer Foundation: Drug Supply: Apotex GCIG Groups: ANZGOG: K. Phillips ISGO: J. Korach, D. Tsoref NCRI: R. Manchanda
Stratification
- Age (<40 vs 40-50 vs >50)
- Current (last dose <6 mths) combined oral
contraceptive use (Yes vs No)
- BRCA status (BRCA1 vs BRCA2)