SLIDE 1
Ring the hydrogen bells! H c H e H g H c H e H g frequency H c H e H - - PowerPoint PPT Presentation
Ring the hydrogen bells! H c H e H g H c H e H g frequency H c H e H - - PowerPoint PPT Presentation
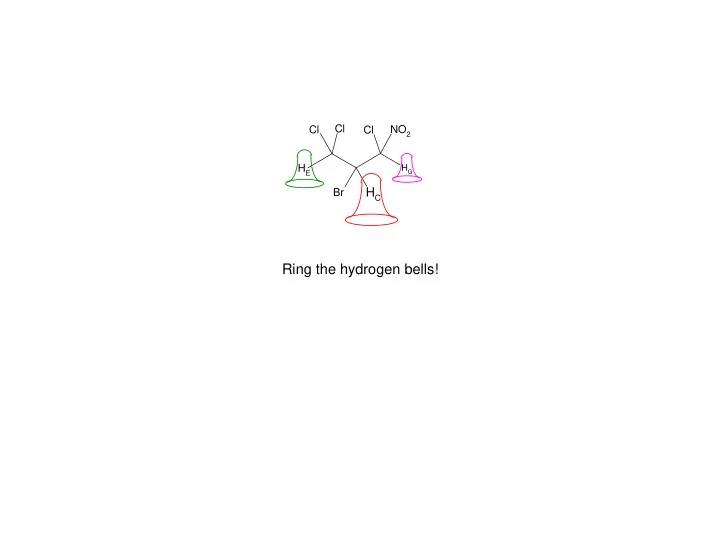
Cl Cl NO 2 Cl H E H G H C Br Ring the hydrogen bells! H c H e H g H c H e H g frequency H c H e H g time frequency time Cl NO 2 Cl Cl H E H G Br H C How do 1 H nuclei ring (or resonate)? the force required to cause ringing is
SLIDE 2
SLIDE 3
frequency time
SLIDE 4
HE HG Cl Br HC Cl Cl NO2
How do 1H nuclei ring (or “resonate”)?
- the force required to cause ringing is radio wave fequency electromagnetic radiation
- the 1H nuclei only ring when placed in a very strong magnetic field
- the pitch (frequency) of the 1H nuclei when ringing is
- proportional to the magnetic field strength
- proportional to the electronic environment of the molecule
SLIDE 5
Nuclear Magnetic Resonance (NMR) Spectroscopy: A kind of emission spectrometry
EM radiation source sampleO sample* sampleO detector, amplifier, & recorder excitation relaxation
in NMR,
- 1. EM source is a radio transmitter, typically in the range of ~60 - 750 MHz
(compare with the FM radio band 88 - 108 MHz)
- 2. Sample holder is a magnet with a test tube in it
- 3. Excitation occurs by absorption of Rf radiation, causing a net change in nuclear spin (“bulk
magnetization”)
- 4. Relaxation occurs by Rf emission and a return of the bulk magnetization to equillibrium
- 5. Detector is a radio receiver
SLIDE 6
Block diagram of an NMR instrument:
SLIDE 7
NMR magnet size: old and new
2001: Scripps superconducting 21.1 Tesla electromagnet, 17' 4" tall. Contains ~ 27 MJ of stored energy 1968: Creighton T60 with 1 Tesla permanent iron core magnet, ~ 18" tall
SLIDE 8
A typical 300 MHz NMR instrument:
SLIDE 9
Cl C Cl H Cl
2477.32 MHz
Chloroform + 0.03% TMS 299.95 MHz
SLIDE 10
The frequency of resonance,
- , of a hydrogen in a molecule is a function of the intrinsic
magnetic properties (the magnetogyric ratio,
- ) of the hydrogen nucleus and the magnetic field,
Bo, that the nucleus feels:
- = (
- Bo) / 2
- To make the frequency of resonance magnetic-field independent, we use a relative scale
called chemical shift which is measured in ppm or
- :
- sample -
- TMS
- = _________
- TMS
In our case, (2477.32 x 106) - (299.95 x 106)
- = _________________________ = 7.26 ppm
299.95 x 106
SLIDE 11
downfield upfield deshielded shielded higher frequency lower frequency
SLIDE 12
(2477.32 MHz) Beff CHCl3 = B0 + BH - Be
CHCl 3
Beff TMS = B0 + BH - Be
TMS
As TMS H’s are more electron rich than the CHCl3 H, Be
TMS > Be CHCl 3 .
So, Beff TMS < Beff CHCl3 and therefore νTMS < νCHCl3 . (299.95 MHz) downfield upfield deshielded shielded higher frequency lower frequency Cl C Cl H Cl H3C Si CH3 CH3 CH3
SLIDE 13
The chemical shift reflects the electronic environment CHCl3 δ 7.3 ppm CH2Cl2 δ 5.2 ppm CH3Cl δ 3.1 ppm
SLIDE 14
The chemical shift reflects the electronic environment CH3I δ 2.2 ppm CH3Br δ 2.7 ppm CH3Cl δ 3.1 ppm
SLIDE 15
H C O H H H
Methanol 1H NMR spectrum
SLIDE 16
H C O H H H
Methanol 1H NMR spectrum Relative ratio of areas: 70 / 23 = 3.0 23 / 23 = 1 3H 1H
SLIDE 17
H C O H H H
Methanol 1H NMR spectrum
SLIDE 18
CH3 CH3 H H H H
para-Xylene 1H NMR spectrum
SLIDE 19
BASIC 1H NMR CHEMICAL SHIFTS (R = H or Alkyl) H Type Approximate δ, ppm methyl 0.9 methylene 1.3 methine 1.5 vinylic R2C=CHR 4.6-5.9 acetylenic RCC–H 2-3 ayllic R2C=CRCHR2 1.7 aryl Ar–H 6.5-8.0 benzylic Ar–CHR2 2.2-3 Cl–CHR2 3-4 Br–CHR2 2.5-4 I–CHR2 2-4 RO–CHR2 3.3-4 RCO2–CHR2 3.7-4.1 O
- R3C–C–CHR2
2-2.6 O
- 9-10
R–C–H O
- 10.5-13