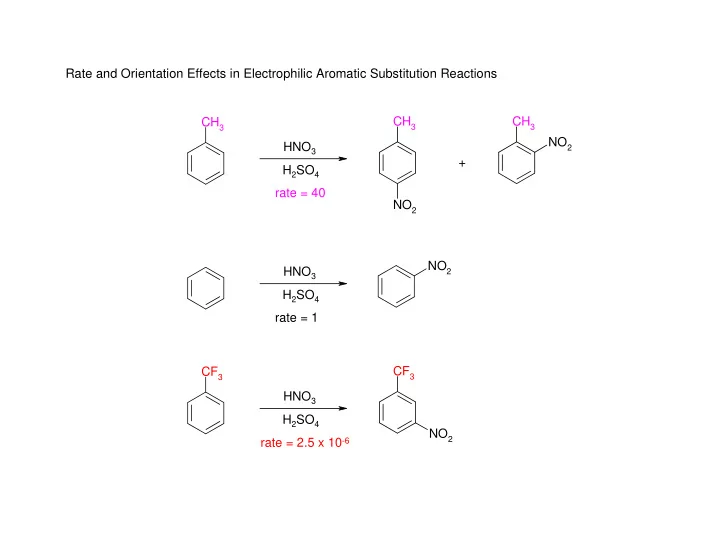
CH3 CH3 NO2 CH3 NO2 NO2 CF3 CF3 NO2 HNO3 H2SO4 rate = 40 + HNO3 H2SO4 rate = 1 HNO3 H2SO4 rate = 2.5 x 10-6 Rate and Orientation Effects in Electrophilic Aromatic Substitution Reactions
Rate and Orientation Effects in Electrophilic Aromatic Substitution - - PowerPoint PPT Presentation
Rate and Orientation Effects in Electrophilic Aromatic Substitution - - PowerPoint PPT Presentation
Rate and Orientation Effects in Electrophilic Aromatic Substitution Reactions CH 3 CH 3 CH 3 NO 2 HNO 3 + H 2 SO 4 rate = 40 NO 2 NO 2 HNO 3 H 2 SO 4 rate = 1 CF 3 CF 3 HNO 3 H 2 SO 4 NO 2 rate = 2.5 x 10 -6 Activating Substituents Deactivating
NH2 .. X .. .. : OH .. .. S OH O O O R N H .. OH O OR O , OR .. .. O R N
+
O O X X X
Activating Substituents Deactivating Substituents
- rtho, para
meta
- rtho, para
–CN –R
Nitration of anisole The resonance donating effects of the OCH3 substituent make it an activating substituent
- The aromatic ring is more nucleophilic than benzene
- The resonance delocalization of the electron density of the aromatic ring is lumpy:
There is greater electron density at the o- and p- carbons
O O O O + . . .. ..
- +
. .
- +
.. -
Nitration of anisole:
- rtho-attack of nitronium ion
O O N
+ O
O H NO2 O H NO2 O H NO2 O
+
H NO2 + + + +
O O N
+ O
O H NO2 O H NO2 O H NO2 O
+
H NO2 + + + + O O N
+ O
O H NO2 O H NO2 O H NO2 + + + +
Nitration of anisole:
- rtho-attack of nitronium ion
meta-attack of nitronium ion
O O N
+ O
O H NO2 O H NO2 O H NO2 O
+
H NO2 + + + + O O N
+ O
O H NO2 O H NO2 O H NO2 + + + + O O N
+ O
O H NO2 O H NO2 O
+
H NO2 O H NO2 + + + +
Nitration of anisole:
- rtho-attack of nitronium ion
meta-attack of nitronium ion para-attack of nitronium ion
O O N
+ O
O H NO2 O H NO2 O H NO2 O
+
H NO2 + + + + O O N
+ O
O H NO2 O H NO2 O H NO2 + + + + O O N
+ O
O H NO2 O H NO2 O
+
H NO2 O H NO2 + + + +
Nitration of anisole:
- rtho-attack of nitronium ion
meta-attack of nitronium ion all octets filled! para-attack of nitronium ion all octets filled!
CH3O CH3O O2N CH3O NO2 CH3O NO2 + +NO2
- - and p-arenium
ions + m-arenium ion
CH3O CH3O O2N CH3O NO2 CH3O NO2 + +NO2
- - and p-arenium
ions + m-arenium ion
Where would the E vs reaction coordinate diagram of the nitration of benzene be placed relative to this one?
Nitration of nitrobenzene The inductive and resonance withdrawing effects of the NO2 substituent make it a deactivating substituent
- The aromatic ring is less nucleophilic than benzene
- The resonance delocalization of the electron density of the aromatic ring is lumpy:
There is greater electron density at the m- carbons
N O O N O O N O O N O O + + + + + + +
N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + +
Nitration of nitrobenzene:
- rtho-attack of nitronium ion
N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + +
Nitration of nitrobenzene:
- rtho-attack of nitronium ion
meta-attack of nitronium ion
N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + +
N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + +
Nitration of nitrobenzene:
- rtho-attack of nitronium ion
meta-attack of nitronium ion para-attack of nitronium ion
N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + + N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + +
N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + + N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + + N O O O N
+ O
N O H NO2 O N O H NO2 O N O H NO2 O + + + + + + + +
Nitration of nitrobenzene:
- rtho-attack of nitronium ion
- meta-attack of nitronium ion
para-attack of nitronium ion
O2N O2N O2N NO2 O2N NO2 O2N + +NO2
- - and p-arenium
ions + m-arenium ion
O2N O2N O2N NO2 O2N NO2 O2N + +NO2
- - and p-arenium
ions + m-arenium ion
Where would the E vs reaction coordinate diagram of the nitration of benzene be placed relative to this one?
Use the same method of analysis to explain why the Cl substituent is a deactivating, o-, p-directing substituent:
Cl Cl NO2 Cl O2N 1:1 HNO3 : H2SO4 +