SLIDE 1
1
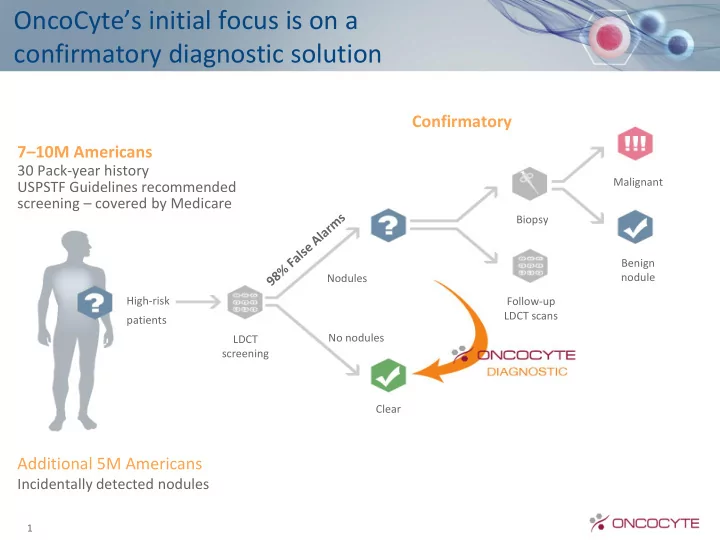
High-risk patients LDCT screening Clear Biopsy Malignant Benign nodule Follow-up LDCT scans Nodules
Confirmatory
OncoCyte’s initial focus is on a confirmatory diagnostic solution
No nodules
OncoCytes initial focus is on a confirmatory diagnostic solution - - PowerPoint PPT Presentation
OncoCytes initial focus is on a confirmatory diagnostic solution Confirmatory 7 10M Americans 30 Pack-year history Malignant USPSTF Guidelines recommended screening covered by Medicare Biopsy Benign Nodules nodule High-risk
1
High-risk patients LDCT screening Clear Biopsy Malignant Benign nodule Follow-up LDCT scans Nodules
No nodules
2
3
4
Patients undergoing biopsy Screen 1,310 protein analytes in multiplexed format Analysis and algorithm development
5