SLIDE 1
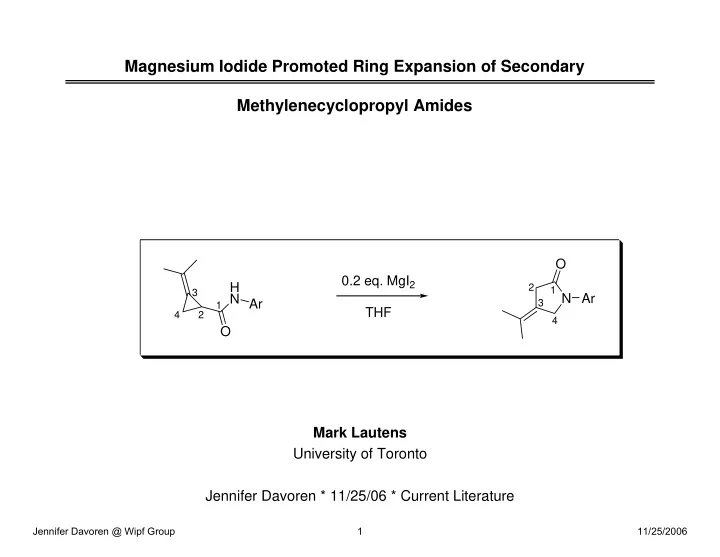
Magnesium Iodide Promoted Ring Expansion of Secondary Methylenecyclopropyl Amides
Mark Lautens University of Toronto Jennifer Davoren * 11/25/06 * Current Literature
H N O Ar N Ar O
1 1 2 2 3 3 4 4
0.2 eq. MgI2 THF
Jennifer Davoren @ Wipf Group 1 11/25/2006

SLIDE 2 Methylenecyclopropane Derivatives
NH2 CO2H CO2H NH2 H hypoglycine methylenecyclopropylglycine methylenecyclopropane
- Hypoglycine is a naturally derived amino acid unripe fruit of
the ackee tree Blighia sapida
- Responsible for Jamaican vomiting sickness
- Methylenecyclopropylglycine was isolated from the kernels of
litchi fruits
- Causes hypoglycemia in mice and fasted rats
- Methylenecyclopropane is a stable volatile olefin (bp 11 °C),
can be stored in a sealed tube for several years without decomposition
Jennifer Davoren @ Wipf Group 2 11/25/2006

SLIDE 3
Synthesis of Methylenecyclopropane Derivatives
Carbene Additions to Allenes NH2 CO2H hypoglycine NH2 CO2H C CH2I2, Zn-Cu 71% The formation of spiropentane derivatives is general and cannot normally be avoided, especially in simple monosubstituted allenes, even when using only a slight excess of the Simmons-Smith reagent NH2 CO2H Eliminations Cl NaNH2 KNH2 The base counterion plays an important role in the nature of products formed, as NaNH2 gives the cyclopropene derivative, whereas KNH2 gives MCP Bn ArSO3 Bu3ZnLi 93% R Bn A nucleophilic alkene can be produced from an alkyne by the addition of an organometallic reagent
Jennifer Davoren @ Wipf Group 3 11/25/2006

SLIDE 4 Synthesis of Methylenecyclopropane Derivatives
Elimination of N2 from Pyrazolines N N H OCOEt H OCOEt OCOEt hv 2:1 H OCOEt H OCOEt R' R OH OEt PPh3 Wittig Olefinations The route employing cyclopropylidene phosphorane has been the most utilized by researchers, because of the unavailability
- f cyclopropanone and the low reactivity of its synthetic
equivalent cyclopropanone hemiacetal
Jennifer Davoren @ Wipf Group 4 11/25/2006

SLIDE 5 [3 + 2] Cycloaddition of Dipolar Trimethylenemethane (TMM) Derived From Methylenecyclopropanes
- J. Org. Chem. 1998, 63, 1694-1703
O O Cl Cl O OH OH H+ Cl Cl O O
- 1. 3 eq NaNH2
- 2. MeI, 76%
O O
δ+ δ-
100 oC CH3CN
N Cl OBn
O O
N OBn Cl
O O
N OBn Cl 99%
Jennifer Davoren @ Wipf Group 5 11/25/2006

SLIDE 6 Carriera’s Precedence
- Angew. Chem., Int. Ed. 1999, 38, 3186
Jennifer Davoren @ Wipf Group 6 11/25/2006

SLIDE 7 Lauten’s Early Work
- J. Am. Chem. Soc. 2002. 124, 6312
- Studies began with the reactions of several monoactivated MCP’s of types 1a-1c with aryl
aldimines in the presence of stoichiometric MgI2
- Reactions using ester 1a and stoichiometric MgI2 recovered starting material
- Whereas amides 1b and 1c gave complex mixtures even in the reactions with aryl
aldehydes
- In contrast the diphenyl amide 1d could be reacted with a variety of imines in good
yields
Jennifer Davoren @ Wipf Group 7 11/25/2006

SLIDE 8 Reactions of MCP Amides: Bearing a Diphenyl Amide
- In the case of aldimines bearing an
- rtho-substituent (entries 7-10) only the
trans diastereomers were obtained.
- Stoichiometric MgI2 is not required, the
reactions could be carried out with 10-30 mol % of MgI2 without any loss in yield (entries 3, 6, and 8-10).
- When 10 mol % MgI2 was used, an
increase in the reaction concentration to 0.2 M was required to ensure complete reaction (entry 6).
- Reactions with aryl iodides provided
complex mixtures of products
Jennifer Davoren @ Wipf Group 8 11/25/2006

SLIDE 9 Reactions of MCP Imides: Bearing a Oxazolidinone
- In contrast to the results with the
diphenyl amide, the products were exclusively six-membered heterocycles bearing an allyl iodide and lacking the oxazolidone group
- Requires a stoichiometric amount
- f MgI2 to go to completion
- The iodo-substituted products were
not stable to silica flash chromatography
- A concerted [4+2] hetero- Diels-
Alder reaction pathway could not be ruled out
Jennifer Davoren @ Wipf Group 9 11/25/2006

SLIDE 10 Reactions of MCP Amides: Bearing a Secondary Amide
- J. Am. Chem. Soc. 2003, 125, 4028.
- A different process for secondary
MCP amides was observed
- In the absence of an electrophile
underwent ring expansion to the isomeric five-membered unsaturated lactam 7
- In the presence of a wide range of
aryl aldimines or aldehydes products such as 8 were obtained
Jennifer Davoren @ Wipf Group 10 11/25/2006

SLIDE 11
Reactions of MCP Amides: Bearing a Secondary Amide
In each case, the monoalkylated product 7 was isolated in 10-30% yield
Jennifer Davoren @ Wipf Group 11 11/25/2006

SLIDE 12 Diastereoselective Ring Expansion of MCP
Jennifer Davoren @ Wipf Group 12 11/25/2006

SLIDE 13 Diastereoselective Ring Expansion of MCP
- For the pyridyl series (entries 1-2), the diastereoselectivity was found to be excellent
in all cases.
- In the furyl series, however, the diastereoselectivity decreased when the oxygen was
- rtho to the imine substituent
Jennifer Davoren @ Wipf Group 13 11/25/2006

SLIDE 14 Proposed Mechanism of Diastereoselective MCP Ring Expansion
- The enolate must attack the sulfinimine
via a boat TS to give the observed anti relationship
- The sulfoxide adopts a conformation in
this boat transition state to minimize 1,3- allylic strain while maximizing the stabilization of this intermediate via coordination of the magnesium to the
- xygen of the sulfoxide.
- Presumably the presence of an ortho
heteroatom in the sulfinimine results in low diastereoselectivity due to competing coordination of magnesium to the ortho heteroatom
products could be deprotected in 94% using TFA
Jennifer Davoren @ Wipf Group 14 11/25/2006

SLIDE 15 Synthesis of β,γ-Unsaturated Lactams via a MgI2 Promoted Ring Expansion of Secondary MCP Amides
- Org. Lett. 2006, 8, 5521
- Initial investigations established that THF
and MgI2 were optimal as both solvent and Lewis acid
- The use of dilute reaction conditions and
substoichiometric amounts of MgI2 were crucial to obtaining the exo isomer in excellent yield and selectivity
Jennifer Davoren @ Wipf Group 15 11/25/2006

SLIDE 16 Scope of MgI2 Promoted Ring Expansion of Secondary MCP Amides
substituted azoles (1-3) afforded the corresponding ring-expanded products in excellent yield and selectivity
- The use of an analogous isoxazole substrate bearing
an oxygen adjacent to the amido functionality resulted in no observable ring expansion (4)
- This result suggests that a nitrogen atom
adjacent to the amido functionality is crucial to
product in good selectivity and yield
- Mild electron withdrawing groups and electron rich
groups gave ring expanded products in excellent yields and selectivities
- Conversely electron withdrawing groups gave
poor exo-selectivities (5 & 10)
- Interestingly, MCPs substituted at either the exo
methylene or cyclopropyl carbon also provided ring- expanded products in moderate to good yields with high selectivities of the exo product
Jennifer Davoren @ Wipf Group 16 11/25/2006

SLIDE 17
Proposed Mechanism of MgI2 Promoted Ring Expansion
Jennifer Davoren @ Wipf Group 17 11/25/2006

SLIDE 18 Conclusions
- MgI2 promoted ring expansion of MCP’s to either 5 or 6 membered rings
- Unique mechanistic pathways observed depending on the type of amide used; ie.
diphenyl amides vs. secondary amides vs. oxazolidinones
- Diastereoselective variant was developed using the diphenyl amide
Jennifer Davoren @ Wipf Group 18 11/25/2006