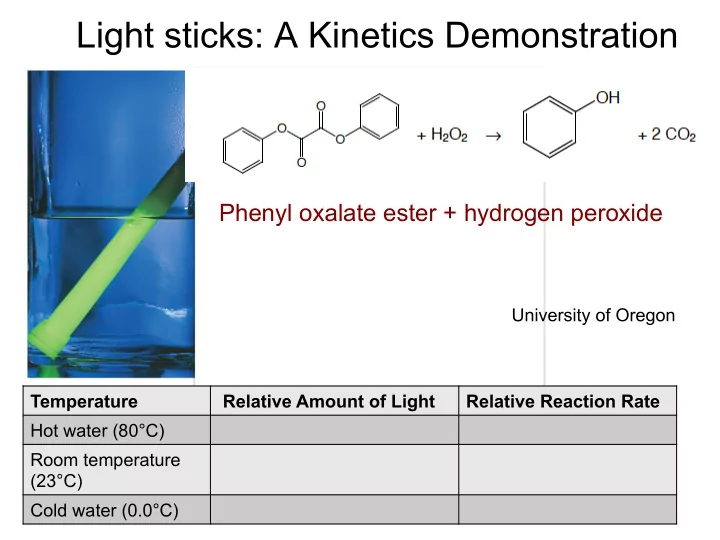
Light sticks: A Kinetics Demonstration
Temperature Relative Amount of Light Relative Reaction Rate Hot water (80°C) Room temperature (23°C) Cold water (0.0°C)
Phenyl oxalate ester + hydrogen peroxide
University of Oregon
Light sticks: A Kinetics Demonstration Phenyl oxalate ester + - - PowerPoint PPT Presentation
Light sticks: A Kinetics Demonstration Phenyl oxalate ester + hydrogen peroxide University of Oregon Temperature Relative Amount of Light Relative Reaction Rate Hot water (80C) Room temperature (23C) Cold water (0.0C) Light sticks: A
Temperature Relative Amount of Light Relative Reaction Rate Hot water (80°C) Room temperature (23°C) Cold water (0.0°C)
University of Oregon
Temperature Relative Amount of Light Relative Reaction Rate Hot water (80°C) Bright Room temperature (23°C) Medium Cold water (0.0°C) Dim
Temperature Relative Amount of Light Relative Reaction Rate Hot water (80°C) Bright Fast Room temperature (23°C) Medium Medium Cold water (0.0°C) Dim Slow
When the temperature increases, the molecular speed __________ . When the molecular speed increases, the number of collisions ________ . Therefore, the rate of reaction __________ when the temperature increases. increases increases increases
(always) However, there is more to it than that.
Minimum energy needed to
T2 > T1 A larger fraction of molecules reacts at a higher temperature.
Minimum energy needed to
T2 > T1 A larger fraction of molecules reacts at a higher temperature.