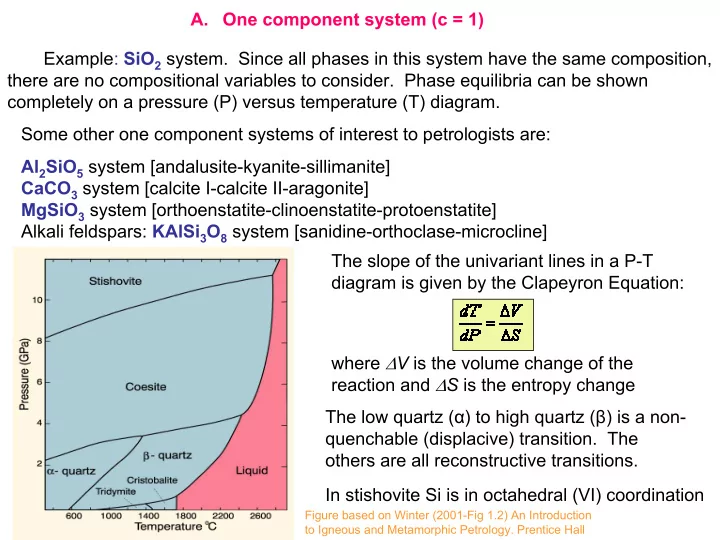
SLIDE 1 Example: SiO2 system. Since all phases in this system have the same composition, there are no compositional variables to consider. Phase equilibria can be shown completely on a pressure (P) versus temperature (T) diagram. where ∆V is the volume change of the reaction and ∆S is the entropy change The slope of the univariant lines in a P-T diagram is given by the Clapeyron Equation: Some other one component systems of interest to petrologists are: Al2SiO5 system [andalusite-kyanite-sillimanite] CaCO3 system [calcite I-calcite II-aragonite] MgSiO3 system [orthoenstatite-clinoenstatite-protoenstatite] Alkali feldspars: KAlSi3O8 system [sanidine-orthoclase-microcline] The low quartz (α) to high quartz (β) is a non- quenchable (displacive) transition. The
- thers are all reconstructive transitions.
In stishovite Si is in octahedral (VI) coordination
- A. One component system (c = 1)
Figure based on Winter (2001-Fig 1.2) An Introduction to Igneous and Metamorphic Petrology. Prentice Hall

SLIDE 2
Stability fields for Al2SiO5 polymorphs
1 2 3 4 5 6 7 8 9 10 200 400 600 800 1000
T (deg C) P (kbars)
S A K
S T V P Path 1 Path 2
Insets show the change in entropy (S) as a function of T along isobaric path 1 and the change in volume (V) along isothermal path 2 (note discontinuities In S and V)

SLIDE 3
- B. Two component systems (binary systems): common types
[1] eutectic system, e.g., Diopside (Di)--Anorthite (An) in which there is little or no solid solution between the end member mineral components [2] peritectic system (with intermediate compound), e.g., Forsterite (Fo)--Silica (SiO2), in which the intermediate compound Enstatite (En) melts incongruently [3] “Double” eutectic system, e.g. Nepheline (Ne)--Silica (SiO2) in which the intermediate compound (Ab) melts congruently. [4] system showing complete miscibility in both liquid and solid phases, e.g., Forsterite (Fo)—Fayalite (Fa) and Albite (Ab)—Anorthite (An) [5] system with solvus, e.g., Albite (Ab)—Orthoclase (Or): Enstatite (En)–Diopside (Di). Such systems exhibit variable solid solution between the end member minerals. [6] System with H2O as a component, e.g., Albite (Ab)—H2O, Quartz—H2O In the first five cases, it is convenient at first to express the equilibria at constant pressure; the phase diagram is referred to as a (TX)P section. In the case where one variable is held constant, the phase rule becomes: f = c - p + 1. Compositional variations are expressed as weight % although we could have expressed the composition in mole %, or in oxygen units.

SLIDE 4 Diopside (CaMgSi2O6)-anorthite (CaAl2Si2O8) system (c = 2)
solidus
P = 1 atm
- 1. Discuss: Cooling of bulk compositions “a” starting at 1450ºC, comp “b” starting at 1600ºC
- 2. Discuss: Melting of compositions “a” and “b” starting at a temperature below the solidus
Concepts: Invariant, univariant and divariant assemblages Equilibrium crystallization and fractional crystallization Equilibrium melting and fractional melting Lever Rule, tie lines Effect of pressure, effect of addition of water to system
a b
Figure from Winter (2001) An Introduction to Igneous and Metamorphic Petrology.

SLIDE 5 In this photomicrograph of a Mount Baker andesite, we would interpret the texture as indicating that plagioclase crystallized at the same time as augite—both are
- euhedral. The texture is “porphyritic.”
In this photomicrograph of a troctolite from the Stillwater Complex, we would interpret the texture as indicating that
- livine crystallized before plagioclase.
“Cumulate” texture.
Examples of crystallization of pyroxene and plagioclase
Plag pyroxene

SLIDE 6
Diopside (CaMgSi2O6)-anorthite (CaAl2Si2O8) system (c = 2): effect of P
40 50 60 P (Gpa) An % 0.5 1 Eutectic composition Note: The effect of increasing pressure is (1) increase the liquidus and solidus T, particularly for Diopside; (2) Change the composition of the eutectic.

SLIDE 7 Diopside (CaMgSi2O6)-anorthite (CaAl2Si2O8) system (c = 2): effect of H2O
Note: Effect of H2O is to:
- 1. Lower melting T dramatically
- 2. Effect of H2O is much more
pronounced in An than in Di, i.e, more polymerized melts show stronger effect.
- 3. Dramatic shift in composition of
the eutectic with H2O saturated conditions.
- 4. Reduced stability of plagioclase
with increasing fH2O T(ºC) T(ºC) P = PH2O (Gpa) 1
1520 1270 1391 1560 1570 1125
Di + V An + V L L L L An Di
1100 1300 1500

SLIDE 8
Binary systems (c = 2) showing complete miscibility in liquid and solid phases
Albite (Ab)—Anorthite (An) Forsterite (Fo)—Fayalite (Fa)
P = 1 atm P = 1 atm
Variables: T, P
L An
X
xal Ab
X
xal An
X
L Ab
X
Phase Rule: f = c – p + 1 = 3 - p Above liquidus: f = ?, Below solidus, f = ? Between liquidus and solidus, f = ?
L Fa
X
xal Fa
X
L Fo
X
xal Fo
X
T, P Phase Rule: f = c – p + 1 = 3 - p Above liquidus: f = ?, Below solidus, f = ? Between liquidus and solidus, f = ?

SLIDE 9
Equilibrium crystallization/melting of composition An60
1500ºC: a=liq, p=1, f =2 1480ºC: liq=b: xals=c, p=2, f=1 1450ºC: liq=d: xals=c: p=2, f=1 1340ºC: liq=g: xals=h: p=2, f=1 <1340ºC: xals=i (An60), p=1, f=2
Tie lines, e.g., b-c, d-f, g-h: Lever Rule: Liq Xals
d e f
de ef Xals Liq =
Figures from Winter (2001) An Introduction to Igneous and Metamorphic Petrology.