SLIDE 1
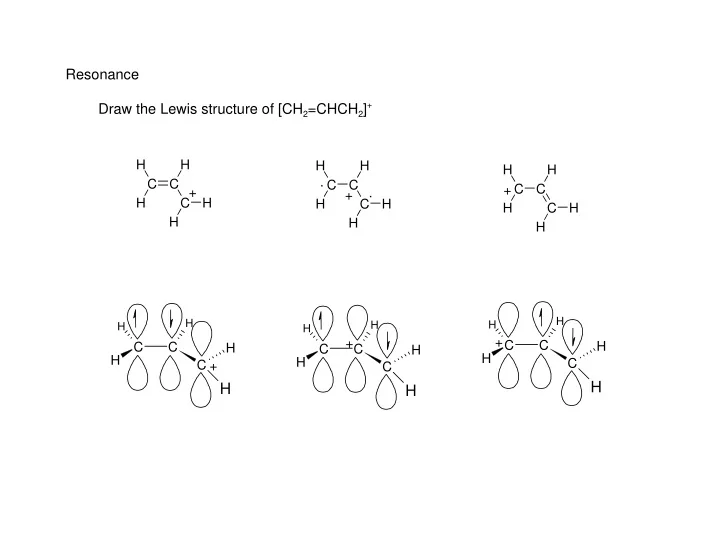
Resonance Draw the Lewis structure of [CH2=CHCH2]+
C C H H H C H H C C H H H C H H C C H H H C H H C C H
H
C
H
H
H
C C H
H
C
H
H
H
C C H
H
C
H
H
H
+ + + . . + + +

SLIDE 2 Resonance Draw the Lewis structure for nitromethane, CH3NO2
N C O O H H H N C O O H H H
+
. . . .. .. . . +
. .. . . . . . .

SLIDE 3
Csp
3
Nsp
2
Osp
2
Osp
2
H1s H1s H1s Csp
3
Nsp
2
Osp
2
Osp
2
H1s H1s H1s
C N O O H H H + .. : : .. .. C N O O H H H + .. : : . .. .

SLIDE 4 Common structural classes where resonance is observed Compounds with allylic lone pairs
O O : : .. -
.. ..

SLIDE 5
Common structural classes where resonance is observed Compounds with allylic positive charge
+ + +

SLIDE 6
Common structural classes where resonance is observed Compounds with lone pairs next to positive charge
Br Br .. .. : + + . .. .

SLIDE 7 Nitromethane, CH3NO2, combines two structural classes where resonance is observed It is a compound with lone pairs next to positive charge and allylic lone pairs
N C O O H H H N C O O H H H
+
. . . .. .. . . +
. .. . . . . . .

SLIDE 8
Common structural classes where resonance is observed Compounds with pi bonds between atoms of different electronegativities
O O . . .. : : .. - +

SLIDE 9
Common structural classes where resonance is observed Compounds with aromatic rings
H H
H H
H H

SLIDE 10 The concepts of resonance
- 1. Resonance is not something that is happening to a molecule. It is a way to describe the electronic
distribution of a molecule.
- 2. Individual resonance isomers are imaginary, not real.
- a. They are representations of a hybrid structure
- b. The resonance hybrid is more stable than any individual resonance isomer contributing to it
- 3. Resonance isomers must be valid Lewis structures.
- 4. Resonance isomers differ only by the relative placement of their pi and nonbonding electrons in p
- rbitals.
- 5. Resonance isomers must have identical constitutions. Therefore, the hybridization states of the
constituent atoms cannot change.
- 6. Resonance isomers must have the same number valence electrons and the same number of
paired and unpaired electrons.
- 7. Different resonance isomers don’t have to be equivalent. Resonance isomers that describe the
electronic configuration of a molecule most realistically have
- a. all octets full
- b. minimal formal charge
- c. minimal formal charge separation
- d. formal charge assignment according to relative electronegativities

SLIDE 11
N H H H H
H
Heterocyclic aromatic compounds: pyridine
N H H H H H N H H H H H N H H H H H .. .. . .- + X

SLIDE 12 Consider resonance isomers of spinacine
.. . .. : O N N N O H H H . . . . . +