More On Carbon Monoxide
− →
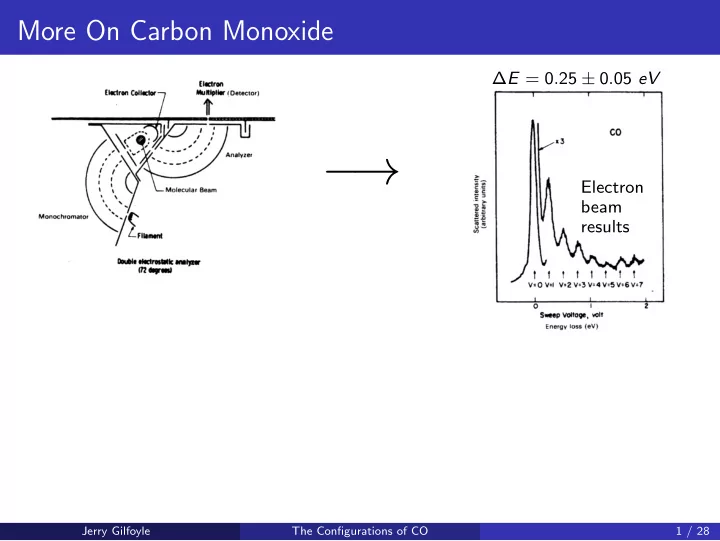
Electron beam results ∆E = 0.25 ± 0.05 eV
Jerry Gilfoyle The Configurations of CO 1 / 28
Electron beam results Jerry Gilfoyle The Configurations of CO - - PowerPoint PPT Presentation
More On Carbon Monoxide E = 0 . 25 0 . 05 eV Electron beam results Jerry Gilfoyle The Configurations of CO 1 / 28 More On Carbon Monoxide E = 0 . 25 0 . 05 eV Electron beam results Jerry Gilfoyle The
Electron beam results ∆E = 0.25 ± 0.05 eV
Jerry Gilfoyle The Configurations of CO 1 / 28
Electron beam results ∆E = 0.25 ± 0.05 eV
Jerry Gilfoyle The Configurations of CO 1 / 28
Electron beam results ∆E = 0.25 ± 0.05 eV
Jerry Gilfoyle The Configurations of CO 1 / 28
Electron beam results ∆E = 0.25 ± 0.05 eV
Incident light CO gas target Photon detector
CO Absorption Spectrum
Jerry Gilfoyle The Configurations of CO 2 / 28
Electron beam results ∆E = 0.25 ± 0.05 eV
Incident light CO gas target Photon detector
CO Absorption Spectrum
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 2 / 28
Excited states of carbon monoxide (CO) can be observed by measuring the absorption spectrum shown below. The molecule can both vibrate and rotate at the same time. The rotational energy states of a rigid rotator are Eℓ = 2 2I ℓ(ℓ + 1) where I is the moment of inertia. The vibrational part of the energy is described by the harmonic oscillator so En = (n + 1
2)ω0 with
∆E = ω0 = 0.25 ± 0.05 eV from our previous results. How do you get the expression above for the rotational energy? Is CO a rigid rotator?
Carbon−Monoxide Spectrum Absorption Energy (eV)
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 3 / 28
Excited states of carbon monoxide (CO) can be observed by measuring the absorption spectrum shown below. The molecule can both vibrate and rotate at the same time. The rotational energy states of a rigid rotator are Eℓ = 2 2I ℓ(ℓ + 1) where I is the moment of inertia. The vibrational part of the energy is described by the harmonic oscillator so En = (n + 1
2)ω0 with
∆E = ω0 = 0.25 ± 0.05 eV from our previous results. How do you get the expression above for the rotational energy? Is CO a rigid rotator?
Carbon−Monoxide Spectrum Absorption Energy (eV)
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 3 / 28
1 What is the kinetic and potential energy between the carbon and
coordinates?
2 How do you decompose the kinetic energy into radial and angular
parts?
3 What is the Schroedinger equation for the rigid rotator? 4 What is the solution of the rigid rotator Schroedinger equation? Jerry Gilfoyle The Configurations of CO 4 / 28
1
2
Jerry Gilfoyle The Configurations of CO 5 / 28
1
2
1
2
2
Jerry Gilfoyle The Configurations of CO 5 / 28
Jerry Gilfoyle The Configurations of CO 6 / 28
Τ
r
α
Jerry Gilfoyle The Configurations of CO 6 / 28
The Laplacian ∇2ψ = 1 r2 ∂ ∂r
∂r
1 r2 sin θ ∂ ∂θ
∂θ
1 r2 sin2 θ ∂2 ∂φ2
Jerry Gilfoyle The Configurations of CO 7 / 28
The Laplacian ∇2ψ = 1 r2 ∂ ∂r
∂r
1 r2 sin θ ∂ ∂θ
∂θ
1 r2 sin2 θ ∂2 ∂φ2
The Schroedinger Equation in 3D − 2 2µ∇2ψ + V (r)ψ = Eψ − 2 2µ 1 r2 ∂ ∂r
∂r
1 r2 sin θ ∂ ∂θ
∂θ
1 r2 sin2 θ ∂2 ∂φ2
+ V (r)ψ = Eψ
Jerry Gilfoyle The Configurations of CO 7 / 28
Steps along the way. − 1 sin θ d dθ
dθ
m2
ℓ
sin2 θΘ = AΘ
Jerry Gilfoyle The Configurations of CO 8 / 28
Steps along the way. − 1 sin θ d dθ
dθ
m2
ℓ
sin2 θΘ = AΘ Legendre’s Associated Equation (1 − z2)d2Θ dz2 − 2z dΘ dz +
m2
ℓ
1 − z2
where z = cos θ
Jerry Gilfoyle The Configurations of CO 8 / 28
Steps along the way. − 1 sin θ d dθ
dθ
m2
ℓ
sin2 θΘ = AΘ Legendre’s Associated Equation (1 − z2)d2Θ dz2 − 2z dΘ dz +
m2
ℓ
1 − z2
where z = cos θ And its recursion relationship when mℓ = 0 ak+2 = k(k + 1) − A (k + 2)(k + 1)ak
Jerry Gilfoyle The Configurations of CO 8 / 28
We have the recursion relationship when mℓ = 0 ak+2 = k(k + 1) − A (k + 2)(k + 1)ak Notice. Given a0 → a2 → a4 · · · and given a1 → a3 → a5 · · · so Θ(z) =
∞
akzk =
∞
akzk +
∞
akzk and we choose a0 = a1 = 1.
Jerry Gilfoyle The Configurations of CO 9 / 28
Red Dashed - ⅇ-ξ2H50
2 (ξ)
Green Dashed - offset × ⅇξ2 ξ log(f(ξ))
ξ = βx β2 = mω0/
Jerry Gilfoyle The Configurations of CO 10 / 28
20 40 60 80 100 0.0 0.5 1.0 1.5 2.0 2.5 kmax Θ(z) Truncated Calculation of Θ(z=1), ml=0
Θ(z = 1) = kmax
k=0 ak(1)k
ak+2 =
k(k+1)−A (k+2)(k+1)ak
Jerry Gilfoyle The Configurations of CO 11 / 28
200 400 600 800 1000 0.0 0.5 1.0 1.5 2.0 2.5 3.0 3.5 kmax Θ(z) Truncated Calculation of Θ(z=1), ml=0
Θ(z = 1) = kmax
k=0 ak(1)k
ak+2 =
k(k+1)−A (k+2)(k+1)ak
Jerry Gilfoyle The Configurations of CO 12 / 28
2000 4000 6000 8000 10000 1 2 3 4 5 kmax Θ(z) Truncated Calculation of Θ(z=1), ml=0
Θ(z = 1) = kmax
k=0 ak(1)k
ak+2 =
k(k+1)−A (k+2)(k+1)ak
Jerry Gilfoyle The Configurations of CO 13 / 28
Blue - Truncated Calculation of Θ(z=1), ml=0 Green - ln(kmax)
2000 4000 6000 8000 10000 1 2 3 4 5 kmax Θ(z) Truncated Calculation of Θ(z=1), ml=0
Θ(z = 1) = kmax
k=0 ak(1)k
ak+2 =
k(k+1)−A (k+2)(k+1)ak
Jerry Gilfoyle The Configurations of CO 14 / 28
ak+2 = k(k + 1) − A (k + 2)(k + 1)ak mℓ = 0 a0 = a1 = 1 Θ = Pℓ(z) =
ℓ
akzk z = cos θ First few polynomials. P0(cos θ) = 1 P3(cos θ) = 1
2
P4(cos θ) = 1
8
2
8
The Configurations of CO 15 / 28
Θ(θ)Φ(φ) = Y m
ℓ (θ, φ) =
4π (ℓ − m)! (ℓ + m)!Pm
ℓ (cos θ)eimφ
Y 0
0 (θ, φ) =
1 √ 4π Y 1
1 (θ, φ) = −
8π sin θeiφ Y −1
1
(θ, φ) =
8π sin θe−iφ Y 0
1 (θ, φ) =
4π cos θ
Jerry Gilfoyle The Configurations of CO 16 / 28
Excited states of carbon monoxide (CO) can be observed by measuring the absorption spectrum shown below. The molecule can both vibrate and rotate at the same time. The rotational energy states of a rigid rotator are Eℓ = 2 2I ℓ(ℓ + 1) where I is the moment of inertia. The vibrational part of the energy is described by the harmonic oscillator so En = (n + 1
2)ω0 with
∆E = ω0 = 0.25 ± 0.05 eV from our previous results. How do you get the expression above for the rotational energy? Is CO a rigid rotator?
Carbon−Monoxide Spectrum Absorption Energy (eV)
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 17 / 28
Excited states of carbon monoxide (CO) can be observed by measuring the absorption spectrum shown below. The molecule can both vibrate and rotate at the same time. The rotational energy states of a rigid rotator are Eℓ = 2 2I ℓ(ℓ + 1) where I is the moment of inertia. The vibrational part of the energy is described by the harmonic oscillator so En = (n + 1
2)ω0 with
∆E = ω0 = 0.25 ± 0.05 eV from our previous results. How do you get the expression above for the rotational energy? Is CO a rigid rotator?
Carbon−Monoxide Spectrum Absorption Energy (eV)
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 17 / 28
p2
r
2µ = − 2 2µ 1 r2 ∂ ∂r
∂r
2µr2 = − 2 2µ
r2 sin θ ∂ ∂θ
∂θ
1 r2 sin2 θ ∂2 ∂φ2
2, ±2 2, ±3 2, ±4 2, ±5 2, ...
sin θ ∂ ∂θ
∂θ
m2
ℓ
sin2 θ
A = ℓ(ℓ + 1) L2|φs = 2ℓ(ℓ + 1)|φs
Jerry Gilfoyle The Configurations of CO 18 / 28
Jerry Gilfoyle The Configurations of CO 19 / 28
Excited states of carbon monoxide (CO) can be observed by passing light through a cell containing CO and measuring the absorption spectrum shown below. The molecule can both vibrate and rotate at the same time. The rotational energy states of a rigid rotator are Eℓ = 2 2I ℓ(ℓ + 1) where I is the moment of inertia. The vibrational part of the energy can is described by the harmonic oscillator so En = (n + 1
2)ω0 with
∆E = ω0 = 0.25 ± 0.05 eV from our previous results. How does one
rotator?
Carbon−Monoxide Spectrum Absorption Energy (eV)
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 20 / 28
Axis at the CM.
Jerry Gilfoyle The Configurations of CO 21 / 28
Axis at the CM.
Jerry Gilfoyle The Configurations of CO 21 / 28
Excited states of carbon monoxide (CO) can be observed by passing light through a cell containing CO and measuring the absorption spectrum shown below. The molecule can both vibrate and rotate at the same time. The rotational energy states of a rigid rotator are Eℓ = 2 2I ℓ(ℓ + 1) where I is the moment of inertia. The vibrational part of the energy is described by the harmonic oscillator so En = (n + 1
2)ω0 with
∆E = ω0 = 0.25 ± 0.05 eV from our previous results. How does one
rotator?
Carbon−Monoxide Spectrum Absorption Energy (eV)
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 22 / 28
Excited states of carbon monoxide (CO) can be observed by passing light through a cell containing CO and measuring the absorption spectrum shown below. The molecule can both vibrate and rotate at the same time. The rotational energy states of a rigid rotator are Eℓ = 2 2I ℓ(ℓ + 1) where I is the moment of inertia. The vibrational part of the energy is described by the harmonic oscillator so En = (n + 1
2)ω0 with
∆E = ω0 = 0.25 ± 0.05 eV from our previous results. How does one
rotator?
Carbon−Monoxide Spectrum Absorption Energy (eV)
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 22 / 28
Electron beam results ∆E = 0.25 ± 0.05 eV
Incident light CO gas target Photon detector
CO Absorption Spectrum
0.2650 0.2600 0.2550 0.2700 0.2750
Jerry Gilfoyle The Configurations of CO 23 / 28
n=0 n=1 n=2
ℓ-value
0 1 2 3 4 5 6 7 8 0 1 2 3 4 5 6 7 8 0 1 2 3 4 5 6 7 8 0.00 0.02 0.04 0.06 0.08 E (eV) CO Rotator Level Scheme
Enl = (n + 1
2)ω0 + 2 2I ℓ(ℓ + 1)
∆En = ω0 = 0.025 ± 0.05 eV ∆Eℓ = 2
I = 0.44 ± 0.07 meV
Jerry Gilfoyle The Configurations of CO 24 / 28
l=5 l=5 n=0 n=1
Jerry Gilfoyle The Configurations of CO 25 / 28
l=5 l=5 n=0 n=1 l=5 n=0 n=1 l=6 l=6 l=7
Vibration−Rotation Adjacent Initial Angular Momenta l+1 l
Jerry Gilfoyle The Configurations of CO 26 / 28
l=5 l=5 n=0 n=1 l=5 l=5 n=0 n=1 l=6 l=6 l=7 l=4
Vibration−Rotation Adjacent Initial Angular Momenta
Jerry Gilfoyle The Configurations of CO 27 / 28
ΔE Probability Rotational Spectra
Jerry Gilfoyle The Configurations of CO 28 / 28