Lecture 14: Anharmonic Oscillator and Raman Effect
Effect Transition Moment Integral Can be evaluated analytically - - PowerPoint PPT Presentation
Effect Transition Moment Integral Can be evaluated analytically - - PowerPoint PPT Presentation
Lecture 14: Anharmonic Oscillator and Raman Effect Transition Moment Integral Can be evaluated analytically Often simplified by symmetry Gives rise to selection rules if recursion formulae exist The chemical bond as a simple harmonic
Transition Moment Integral
- Can be evaluated analytically
- Often simplified by symmetry
- Gives rise to selection rules if recursion formulae exist
The chemical bond as a simple harmonic oscillator SHO: a good approximation for small displacements Parabolic potential: V x
( ) = 1
2 kx2
Schrödinger equation: Boundary condition: = 0 at x = + ∞ Ev = (v + ½)ħω
The chemical bond as a simple harmonic oscillator v = Vibrational quantum number = 0, 1, 2, 3, …
w = k m
Force constant Reduced mass Ev+1 – Ev = Ev=0 = ½ħω Zero point energy = h /2 = hn
n = 1 2p k m n = 1 2pc k m
Spectrum of a harmonic oscillator v = Vibrational quantum number = 0, 1, 2, 3, …
w = k m
Force constant Reduced mass Ev+1 – Ev = = h /2 = hn
n = 1 2p k m n = 1 2pc k m
Energy of transition Intensity
1000 2000 3000
IR Spectrum: Bond strength Polyatomic molecule: Different Bond strengths Functional groups Dv=1
High resolution IR spectrum of HCl Rotational fine structure Isotope effect Dv=1, DJ=+1
Anharmonic oscillator SHO: a good approximation
- nly for small displacements
- The bond breaks at large displacements
- Bond dissociation energy
Morse potential
Anharmonic oscillator
- The bond breaks at large displacements
- Bond dissociation energy
- Energy levels come closer for higher values of v
- Fundamental and overtones in IR spectra
SHO: a good approximation
- nly for small displacements
Morse potential
Anharmonic oscillator: Energies
- The bond breaks at large displacements
- Bond dissociation energy
- Energy levels come closer for higher values of v
- Fundamental and overtones in IR spectra
SHO: a good approximation
- nly for small displacements
ev = (v + ½)n - (v + ½)2xen Morse potential xe = n/4De Dv=+1, +2, +3,….
Anharmonic oscillator: “Selection” rules
- The bond breaks at large displacements
- Bond dissociation energy
- Energy levels come closer for higher values of v
- Fundamental and overtones in IR spectra
ev = (v + ½)n - (v + ½)2xen Morse potential xe = n/4De Dv=+1, +2, +3,…. Fundamental 1st Overtone 2nd Overtone
Anharmonic oscillator: Position of spectral lines ev = (v + ½)n - (v + ½)2xen xe = n/4De Dv=+1, +2, +3,…. Fundamental 1st Overtone 2nd Overtone
e0 = 1 2 n - 1 4 xen e1 = 3 2n - 9 4 xen e2 = 5 2 n - 25 4 xen e3 = 7 2 n - 49 4 xen
Anharmonic oscillator: Position of spectral lines xe = n/4De Dv=+1, +2, +3,…. Fundamental 1st Overtone 2nd Overtone
e0 = 1 2 n - 1 4 xen e1 = 3 2n - 9 4 xen e2 = 5 2 n - 25 4 xen e3 = 7 2 n - 49 4 xen
e1 -e0 =n -2xen =n(1-2xe) e2 -e0 = 2n -6xen = 2n(1-3xe) e3 -e0 = 3n -12xen = 3n(1-4xe)
Anharmonic oscillator: Position of spectral lines xe = n/4De Dv=+1, +2, +3,…. Fundamental 1st Overtone 2nd Overtone
e0 = 1 2 n - 1 4 xen e1 = 3 2n - 9 4 xen e2 = 5 2 n - 25 4 xen e3 = 7 2 n - 49 4 xen
Intensity Wavenumber
e1 -e0 =n -2xen =n(1-2xe) e2 -e0 = 2n -6xen = 2n(1-3xe) e3 -e0 = 3n -12xen = 3n(1-4xe)
IR Spectrum of Carbon Monoxide Fundamental Peak First Overtone 2143 cm-1 4260 cm-1
IR Spectrum of Carbon Monoxide: High resolution Fundamental Peak First Overtone 2143 cm-1 4260 cm-1
Population of states and hot band Morse potential Typical energy gap: 100s and 1000s of cm-1
v
n µ exp -ev / kT
( )
Boltzmann distribution:
v=1
n
v=0
n
= 0.008
for energy gap of 1000 cm-1 High temperature: v =1 to v = 2 ….. are possible Hot band Intensity of hot band: Population of v =1 at that temperature
How to find out bond length of H2 ?
- Polarizability: Induced dipole moment.
- Molecular rotation or vibration: Oscillating induced dipole
- Scattering of (usually) visible monochromatic light by
molecules of a gas, liquid or solid
- Two kinds of scattering :
– Rayleigh (1 in every 10,000) : No change in frequency – Raman (1 in every 10,000,000): Change in frequency Raman Spectroscopy Diatomic molecule, NO permanent dipole moment
Raman scattering: different from original Raman Spectroscopy Rayleigh scattering: no change in energy of light Anti- Stokes shift Stokes shift Virtual level Dn (= n nex) nex n
- Dn : energy gaps in molecule
- Dn : No dpendence on nex
- Stokes strong, anti-Stokes
weak for vibrational levels
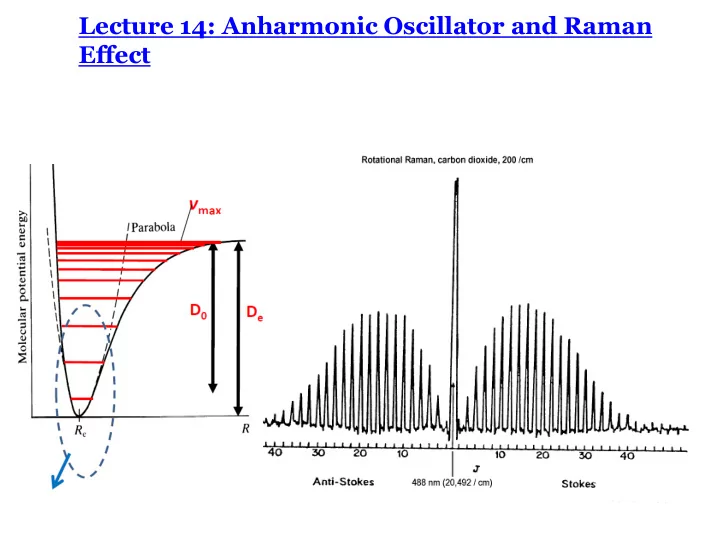
Rotational Raman Spectroscopy: CO2
0, 2 J D =
Selection Rule:
CH 107 in a nutshell: Quantum mechanics in Chemistry: Theory and its manifestations
ˆ Hy = Ey
CH 107 in a nutshell: Bold thoughts from great minds
CH 107 in a nutshell: Don’t be a frog in the well. “Seek, and ye shall find”
history.cultural-china.com vkaisthaaseem.blogspot.com
All the best for all that you do in future