1
4/14/11 1
Class 19 : Where did the elements come from? 4/14/11 1 Notation - - PDF document
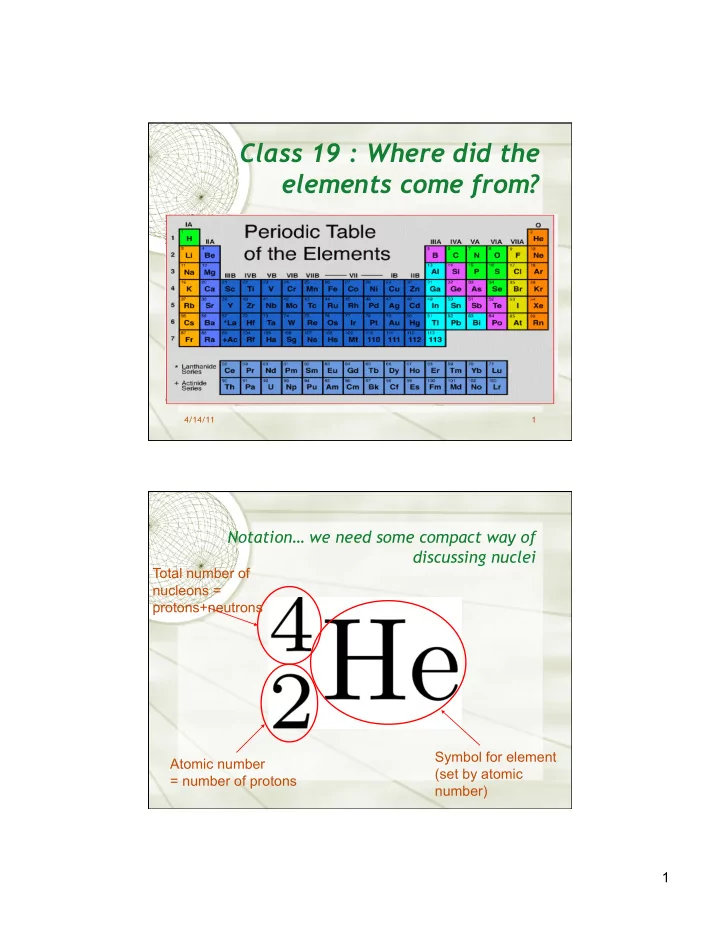
Class 19 : Where did the elements come from? 4/14/11 1 Notation we need some compact way of discussing nuclei Total number of nucleons = protons+neutrons Symbol for element Atomic number (set by atomic = number of protons number) 1
4/14/11 1
4/14/11 3
i.e. 3 minutes after big bang Universe has cooled down to 1 billion (109) K Filled with
Photons (i.e. parcels of electromagnetic radiation) Protons (p) Neutrons (n) Electrons (e) [also Neutrinos, but these were freely streaming]
4/14/11 4
High-T means that photons carry a lot of energy Deuterium is destroyed by energetic photons as
4/14/11 5
e.g. Helium-4 (4He) formation:
4/14/11 6
4/14/11 7
4/14/11 8
If equal number of p and n, everything would
Would have small traces of other species But we know that most of the universe is
4/14/11 9
4/14/11 10
Neutrons spontaneously and randomly decay into
Half life for this occurrence is 10.5 mins (i.e.,
4/14/11 11
Density (affects chance of p/n hitting each other) Temperature (affects how hard they hit) Expansion rate of early universe (affects how
4/14/11 12
Work through all relevant nuclear reactions Take account of decreasing density and
Take account of neutron decay
Turns out that relative elemental abundances
Here, ΩB is the density of the baryons (everything
4/14/11 13
From M.White’s webpage, UC Berkeley
4/14/11 14
These need to be corrected for reactions in stars
4He, and 7Li, we can
Test the consistency of the big bang model -- are
Use the results to measure the quantity ΩBh2
4/14/11 15
All things considered, we
If H0=72km/s/Mpc,
h=0.72 ΩB≈0.04
This is far below Ω=1! Baryons alone would
4/14/11 18
There are naturally occurring elements as heavy
Some elements (e.g., Carbon, Nitrogen, Oxygen)
Astronomers believe these elements were formed
Theory of stellar nucleosynthesis was first worked out
by Burbidge, Burbidge,Folwer, & Hoyle in 1957
4/14/11 19
In the normal life of a
nuclear fusion turns
Hydrogen into Helium
In the late stages of the
Helium converted into
heavier elements (carbon, oxygen, …, iron)
“Triple-alpha” process
bridges stability gap from Be to C
At end of star’s life, get
an onion-like structure (see picture to right)
4/14/11 20
4/14/11 21
Iron has the most stable nucleus Fusing hydrogen to (eventually) iron releases
Further fusion of iron to give heavier elements
Can only happen in the energetic environment of
4/14/11 26
4/14/11 27
The Crab
We directly see
4/14/11 28