CAT-5571 as a novel therapeutic that reduces infection and controls inflammation in cystic fibrosis
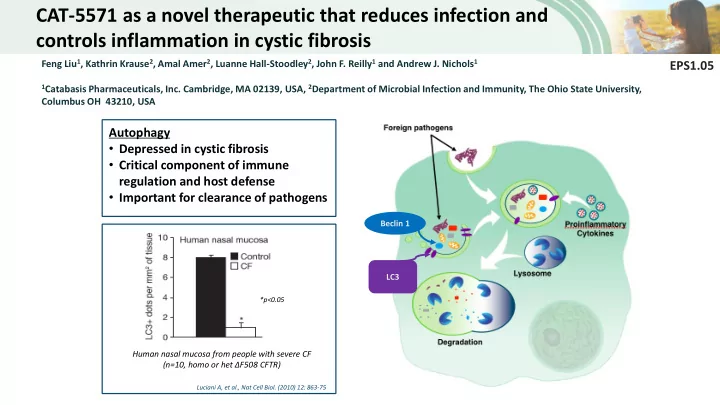
Autophagy
- Depressed in cystic fibrosis
- Critical component of immune
regulation and host defense
- Important for clearance of pathogens
Feng Liu1, Kathrin Krause2, Amal Amer2, Luanne Hall-Stoodley2, John F. Reilly1 and Andrew J. Nichols1
1Catabasis Pharmaceuticals, Inc. Cambridge, MA 02139, USA, 2Department of Microbial Infection and Immunity, The Ohio State University,
Columbus OH 43210, USA
Luciani A, et al., Nat Cell Biol. (2010) 12: 863-75
Human nasal mucosa from people with severe CF (n=10, homo or het ΔF508 CFTR)
*p<0.05
Beclin 1 LC3