5/26/2018 1 Dysplasia in Barrett’s Esophagus and Inflammatory Bowel Disease: Diagnosis, Biomarkers, and Management
Won-Tak Choi, MD, PhD Assistant Professor Department of Pathology University of California San Francisco San Francisco, California
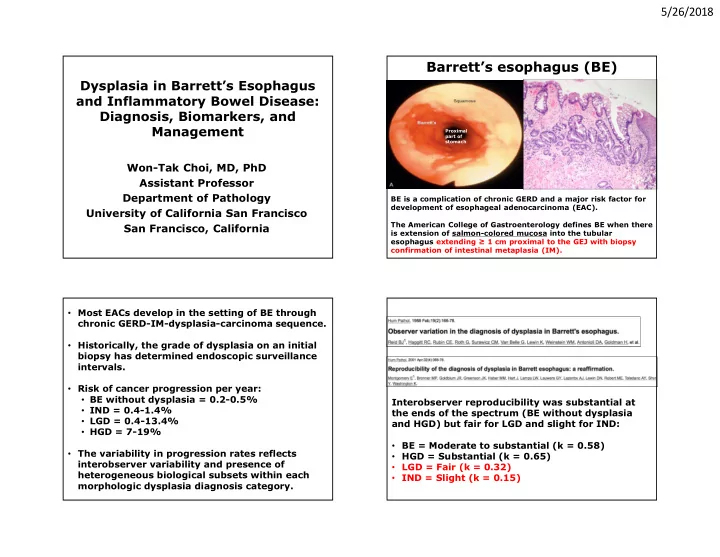
Barrett’s esophagus (BE)
BE is a complication of chronic GERD and a major risk factor for development of esophageal adenocarcinoma (EAC). The American College of Gastroenterology defines BE when there is extension of salmon-colored mucosa into the tubular esophagus extending ≥ 1 cm proximal to the GEJ with biopsy confirmation of intestinal metaplasia (IM).
Distal esophagus Proximal part of stomach
- Most EACs develop in the setting of BE through
chronic GERD-IM-dysplasia-carcinoma sequence.
- Historically, the grade of dysplasia on an initial
biopsy has determined endoscopic surveillance intervals.
- Risk of cancer progression per year:
- BE without dysplasia = 0.2-0.5%
- IND = 0.4-1.4%
- LGD = 0.4-13.4%
- HGD = 7-19%
- The variability in progression rates reflects
interobserver variability and presence of heterogeneous biological subsets within each morphologic dysplasia diagnosis category. Interobserver reproducibility was substantial at the ends of the spectrum (BE without dysplasia and HGD) but fair for LGD and slight for IND:
- BE = Moderate to substantial (k = 0.58)
- HGD = Substantial (k = 0.65)
- LGD = Fair (k = 0.32)
- IND = Slight (k = 0.15)