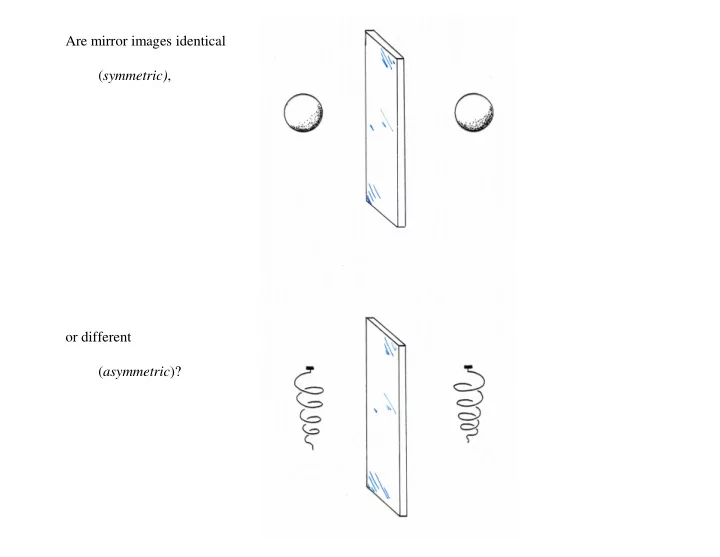
SLIDE 1 Are mirror images identical (symmetric),
(asymmetric)?

SLIDE 2
Left hand mirror
Right hand the left and right hands are nonsuperimposable

SLIDE 3
A molecular case: the amino acid alanine Symmetric or asymmetric?

SLIDE 4 Molecules with the same constitution that differ only by the relative positions of their atoms in space: 1 stereoisomers! Molecular stereoisomers that are nonsuperimposable mirror images are called 1 enantiomers
- An enantiomer possesses the property of molecular chirality.
Enantiomers
- Usually have 4 different functional groups bonded to an sp3-hybridized atom. That atom is
referred to as a 1 Stereogenic center 1 Stereocenter 1 Chiral center
- Have identical physical properites save for one: they cause exactly opposite rotations of
plane-polarized light and so are said to exhibit optical activity. 1 A clockwise rotation is ° (+) ° dextrorotatory ° d 1 A counterclockwise rotation is ° (-) ° levorotatory ° l

SLIDE 5
Light: electromagnetic radiation
Electric
field
Electric
wave
Magnetic
Magnetic wave field
Oscillating orthogonal electric and magnetic fields of a beam of light

SLIDE 6 Plane-polarized light and its consequences
1st polarizing
2nd polarizing filter filter
maximum light source plane- polarized light
no light plane- polarized light

SLIDE 7
Polarimeter: An instrument to measure optical activity

SLIDE 8 [ α ]EC
D = 100 α
cl % e.e. = *[(+)-enantiomer] - [(-)-enantiomer]* x 100 [(+)-enantiomer] + [(-)-enantiomer] % e.e. = α x 100 [ α ]EC
D
Defining a physical constant: Specific rotation " = the observed rotation of plane-polarized light in degrees c = the concentration in g / 100mL l = the pathlength (always 1 dm) Enantiomeric purity (% e.e.): Note: a 50 : 50 mixture of enantiomers has no ". It appears to be achiral. Such mixtures are called racemates.

SLIDE 9 SO WHAT?
Is molecular chiralty important? Consider:
- Gloves
- Smells
- Drug-receptor interactions
Different stereoisomers like enantiomers interact with one another in different ways!

SLIDE 10
Stereochemical theory of odor

SLIDE 11
H O H O H H
(S)-(+)-CARVONE (R)-(-)-CARVONE smells like caraway seeds smells like spearmint gum (R)-(+)-LIMONENE (S)-(-)-LIMONENE smells like citrus fruit smells like turpentine

SLIDE 12 Some drugs that exist as enantiomers and why this matters
(-)-ibuprofen (+)-ibuprofen [α]D
25 = - 20.6E (c 1.00, ethanol)
[α]D
25 = + 58.5E (c 2.00, ethanol)
Advil, Motrin OTC for arthritis, fever; general anti-inflammatory analgesic (+)-enantiomer is the active isomer (-)-enantiomer is inactive the enzyme 2-arylpropionyl-CoA epimerase converts (-)-enantiomer to the (+)-enantiomer
(Zhao, X.; Eur. J. Med. Chem. 2006, 41, 1352-1358; Piccolo, O.; J. Org. Chem. 1985, 50, 3945-3946.)
O OH O O H

SLIDE 13 Some drugs that exist as enantiomers and why this matters
(-)-thalidomide (+)-thalidomide [α]D
25 = - 64.6E (DMF)
[α]D
25 = + 64E (DMF)
teratogen antiemetic enantiomers are interconvertable (undergo racemization) in vivo racemate is immunomodulator used to treat erythema nodosum leprosum (in combination with dexamethasone) newly diagnosed multiple myeloma actinic prurigo
(Ellis, G.P.; West, G.B. Progress in Medicinal Chemistry, Elsevier: New York, 198, p 170) N O H O N O O N O H O N O O

SLIDE 14 Some drugs that exist as enantiomers and why this matters
(-)-ketamine (+)-ketamine [α]D
25 = - 91.88E (c 2.00, H2O)
[α]D
25 = + 92.48E (c 2.00, H2O)
analgesic racemate used to induce general anesthesia (+)-enantiomer (Ketanest S) is more potent (-)-enantiomer is more hallucinogenic: may cause nightmares
(Marrietta, M. P.; Way, W.L.; Castagnoli, N. Jr.; Trevor, A.J. J. Pharmacol. Exp. Ther. 1977, 202, 157-165.) N Cl O H N Cl O H

SLIDE 15
N H N H (+)-methamphetamine (-)-methamphetamine The Alan Baxter story
2002 Salt Lake City Winter Olympics bronze medalist in downhill skiing (slalom) failed IOC routine drug test: + for methamphetamine source of + : Vicks nasal spray U.S. version contains the legal enantiomer of methamphetamine: (decongestant) (crank) IOC test did not differentiate between enantiomers Baxter cleared, but lost his medal – now IOC explicitly bans both enantiomers 2006 Turin Winter Olympics Baxter finishes 16th

SLIDE 16 The R,S system of naming chiral centers:
- Use the Cahn-Ingold-Prelog “Priority Rules”
- 1. Rank the priorities of the 4 atoms attached to the chiral center, according to
atomic number.
- 2. If 2 or more atoms attached are identical, rank the atoms attached to those
atoms, two bonds away from the chiral center. Priority of the entire substituent is determined by the first point of difference. The highest priority substituent is assigned a 1, the next, 2, then 3, and the lowest, 4.
- 3. Position the molecule so that your line of sight is from the chiral center, directly
back to the priority 4 substituent.
- 4. If priority groups 1, 2, and 3 are arranged clockwise in descending rank, the chiral
center is assigned the stereodesignator R
- 4. If priority groups 1, 2, and 3 are arranged counterclockwise in descending rank,
the chiral center is assigned the stereodesignator S

SLIDE 17 C X Y Z C X X C Y Z
For comparison purposes, identical atoms must be compared in the same hybridization state:
- The “Phantom atom” Rules
- 1. For naming purposes, a π bonded atom is considered to replicated by a phantom
σ bond to a phantom atom.
- 2. Rule 1 is applied to both atoms of the π bond.
- 3. Rules 1 and 2 apply to each π bond of multiply bonded atoms.
- 4. All other things being equal, phantom atoms rank below real atoms of the same
atomic number.

SLIDE 18
Cl H An example: Is this an R or an S molecule? So, it is R. Cl H C C Cl H H H H C C C Cl H H H C H H C Cl H 1 4 1 4 3 2

SLIDE 19 What ways are there to draw enantiomers?
- 1. Hatched and wedged line projections (most popular)
are these these enantiomers, or just different hatched and wedged line projections of the same molecule?
- 2. Sawhorse projections could be used, too:
C H3 H H CH3
H H H

SLIDE 20
H O H O H H
Remember carvone and limonene? Do these have R or S configurations?

SLIDE 21 What ways are there to draw enantiomers?
Are these anti-staggered and gauche staggered conformations of the same molecule?
C H3 CH2CH3 H H H CH2CH3 H CH2CH3 CH3 CH3CH2 H H

SLIDE 22 CH3 C C H2 H3C Br H 1 2 3 4 CH3 C CH2 CH3 Br H
What ways are there to draw enantiomers?
\
- a. Orient your point of view so that your body lines up with the parent chain of the molecule and
your line of sight bisects the bond angle formed by the remaining two substituents of the chiral center.
- b. Draw the molecule as it appears to you
using wedged and hatched lines:

SLIDE 23
- c. The Fisher convention replaces wedged and hatched lines with regular ones.
- The vertical lines of a chiral center are always pointing away from you
- The horizontal lines of a chiral center are always pointing towards you
Two of these Fisher projections of different conformations of the same configuration. Which two?
CH3 CH2CH3 Br H CH2CH3 CH2CH2CH2CH3 H O H CH2CH3 H OH CH3CH2CH2CH2 CH2CH3 CH2CH2CH2CH3 OH H

SLIDE 24
Draw (R)-2,3-dimethylheptane as a wedged and hatched line structure and as a Fisher projection:

SLIDE 25 A molecule with 2 chiral centers and 4 stereoisomers 2,3-dihydroxybutanoic acid, also called tartaric acid:
- Draw all four R,S isomers of this molecule
O OH OH OH * *

SLIDE 26
CH3 O OH O H H O H H H3C O O H OH H OH H CH3 O OH H O H O H H H3C O O H H OH OH H CH3 H OH H OH CO2H CH3 H O H H O H CO2H CH3 H OH O H H CO2H CH3 H O H OH H CO2H (2R,3R) (2S,3S) (2R,3S) (2S,3R) enantiomers enantiomers diastereomers
A molecule with 2 chiral centers and 4 stereoisomers

SLIDE 27
OH OH
CH3 CH3 O H H O H H H3C H3C OH H OH H CH3 CH3 H O H O H H H3C H3C H OH OH H CH3 H OH H OH CH3 CH3 H O H H O H CH3 CH3 H OH O H H CH3 CH3 H O H OH H CH3 (2S,3R) = meso (2R,3S) = meso (2S,3S) (2R,3R) the same enantiomers diastereomers diastereomers
A molecule with 2 chiral centers and 3 stereisomers: one isomer is a meso isomer
Consider with 2 chiral centers and (not counting conformational isomers) 22 = 4 possible stereoisomers, but there are only 3!

SLIDE 28
O O OH OH Cl O Cl O H HO OH Cl
O O OH OH OH O O H HO OH OH O H
sucralose (Splenda™) 400-800 times as sweet as sugar sucrose

SLIDE 29
How many chiral centers does Splenda have? nine What is the theoretical maximum number of stereoisomers can this sugar substitute have? 2n , where n = number of stereogenic groups: 29 = 512
O O OH OH Cl O Cl O H HO OH Cl

SLIDE 30
Are the configurations of Splenda’s indicated chiral centers R or S?
O O OH OH Cl O Cl O H HO OH Cl Cl H C C O C H O H C C O C O C C O O Cl
R
1 2 3 4 3 2 4 1
R