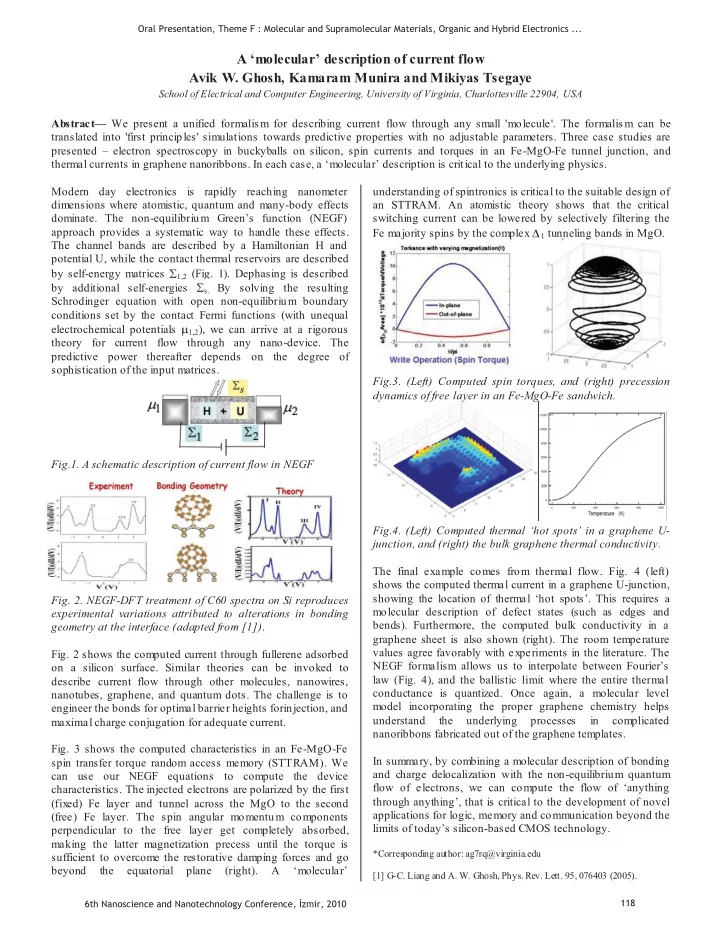
SLIDE 9 Effect of the SAM layer on the Electrical Characteristics and Interface Properties of the ITO/SAM/TPD/Al Schottky Diode
Ali Kemal Havare, 1 Salih Okur, 2* Nesli Yagmurcukardes2, 2, Mustafa Can1 and Serafettin Demic1,
2Izmir Institute of Technology, Faculty of Science, Department of Physics Gülbahce Koyu Kampüsü, Urla, Izmir, 35430, Turkey 1Ege University, Institute of Solar Energy, Bornova, 35100 Izmir, Turkey
Abstract-The electrical characteristics of the ITO/TPD/Al and ITO/SAM (THIBSi)/TPD/Al Schottky diodes have been investigated by current– voltage (I–V) characteristics. We used new SAM molecules to construct ITO/SAM/TPD/Al Schottky devices, and studied the influence on charge tunneling. An ideality factor higher than unity can result from the interface state. The ideality factor n and b
The device performance of a Schottky diode depends on electrical and electronic characteristics of the metal/organic
values of the ITO/TPD/Al and ITO/SAM (THIBSi)/TPD/Al diode were found to be 1.24 and 0.85 eV, 1.28 and 0.40 eV, respectively. The barrier height of the diode was determined from I–V characteristics.
Semiconductor junction [1]. Therefore, the understanding of electronic properties of the interface between metal and
- rganic semiconductors is important for organometalic device
applications [2, 3]. The electronic parameters and interface properties of the ITO/TPD/Al and ITO/SAM (THIBSi)/TPD/Al Schottky diodes have been investigated by current-voltage characteristics. In this study, Schottky energy barrier was constituted between metal and organic material. Schottky contact exhibits a reproducible rectifying behavior and the I–V characteristics are well explained by the conventional Schottky-barrier junction model. The electronic parameters of the diode such as ideality factor and barrier height are compared with ITO/Al, ITO/TPD/Al and ITO/SAM (THIBSi)/TPD/Al. We have synthesized (4'-iodobifenil-4-il) trihidroksisilan (THIBSi) for
- rganic semiconductors as SAM layer. Fig.1 shows the
structure of the SAM molecule and Schottky diode.
Figure 1. Synthesis of SAM molecule and Structure of Schottky diode
Schottky diode was prepared with self-assembly monolayer (SAM) technique. We prepared 1mM THIBSi SAM solutions with chloroform. ITO were kept in the solutions for two days to be formed the SAM film, and then rinsed and dried. After that TPD was evaporated to constitute 50 nm thin film. Finally 200 nm Al was formed as a top contact.
10-7 10-5 0,001 0,1 10 1000
2 4 6
ITO/SAM (THIBSi)/TPD/Al ITO/TPD/Al ITO/Al
Current (mA) Voltage (V)
Figure 2. The I- V characteristic of the ITO/Al Schottky diode.
The I–V characteristics of the ITO/SAM (THIBSi)/TPD/Al Schottky contact are shown in Fig. 2. It shows that the SAM layer enhances the charge tunneling in ITO/SAM
(THIBSi)/TPD/Al
with respect to ITO/TPD/Al diode. Electronic parameters such as ideality factor, barrier height of ITO/SAM (THIBSi)/TPD/Al Schottky junction have been investigated by relation;
qV nkT qV kT q T AA I
b
exp 1 exp exp
2 *
(1) where n is the ideality factor, A is the contact area, T is the temperature, A* is the Richardson constant, q is the electronic charge, k is Boltzmann constant, b
0,05 0,1 0,15 0,2 0,25 0,3 0,35 0,4 0,0005 0,001 0,0015 0,002 0,0025 0,003
dv/d ln(I) ITO/Al dv/dln(I) ITO/TPD dv/dln(I) ITO/SAM/TPD
y = 0,034026 + 125,51x R= 0,99022 y = 0,032279 + 89,818x R= 0,98236 y = 0,033093 + 316,88x R= 0,98793
dV/d lnI (V) Current (A)
is the barrier height.
1 2 3 4 5 0,05 0,1 0,15 0,2 0,25 0,3 0,35
ITO/Al ITO/TPD/Al ITO/SAM(THIBSi)/TPD/Al
y = 0,94931 + 26,668x R= 0,94217 y = 1,0783 + 18,627x R= 0,92937 y = 0,50218 + 37,123x R= 0,9983
H(I) Current Figure 3. The plots of dV/dln (I) - I and H (I) - I of the ITO/Al Schottky diode
The series resistance can be evaluated using a method developed by Cheung [4] to determine the barrier height, ideality factor, and series resistance. Cheung’s functions are defined as,
s
R q kT n
dV
(2)
b s
n IR I H T AA I q kT n V I H
( ) ln( ) (
2 *
(3) The plots of dV/dln (I) - I and H (I) - I are shown in Fig. 3. The diode shows a nonideal current-voltage behavior due to higher than unity. The n values were obtained from the intercept of dV/dln (I) - I plot for ITO/TPD/Al and ITO/SAM
(THIBSi)/TPD/Al, respectively and were found to be 1.24 and
1.28. b and Rs
- values were calculated from the H (I) - I
plot using the obtained were found to be 0.85 eV and 0.40 eV and 18 k 37 k , respectively. This work was supported by TUBITAK under Grant No. TBAG-108T718. *Corresponding author: salihokur@iyte.edu.tr
[1] Willander, M.; Assadi, A.; Svensson, C. Synth. Met. 1993, 55, 4099 [2]Fahrettin YakuphanogluJ. Phys. Chem. C 2007, 111, 1505 1507 [3] F. Yakuphanoglu,S. Okur,H.Özgener Mic. Eng. 86 (2009) 2358 [4] Cheung, S. K.; Cheung, N. W. Appl. Phys. Lett. 1986, 49, 85.
ITO SAM TPD Al Oral Presentation, Theme F : Molecular and Supramolecular Materials, Organic and Hybrid Electronics ... 6th Nanoscience and Nanotechnology Conference, zmir, 2010 126