A brief summary of isotopes and fractionation
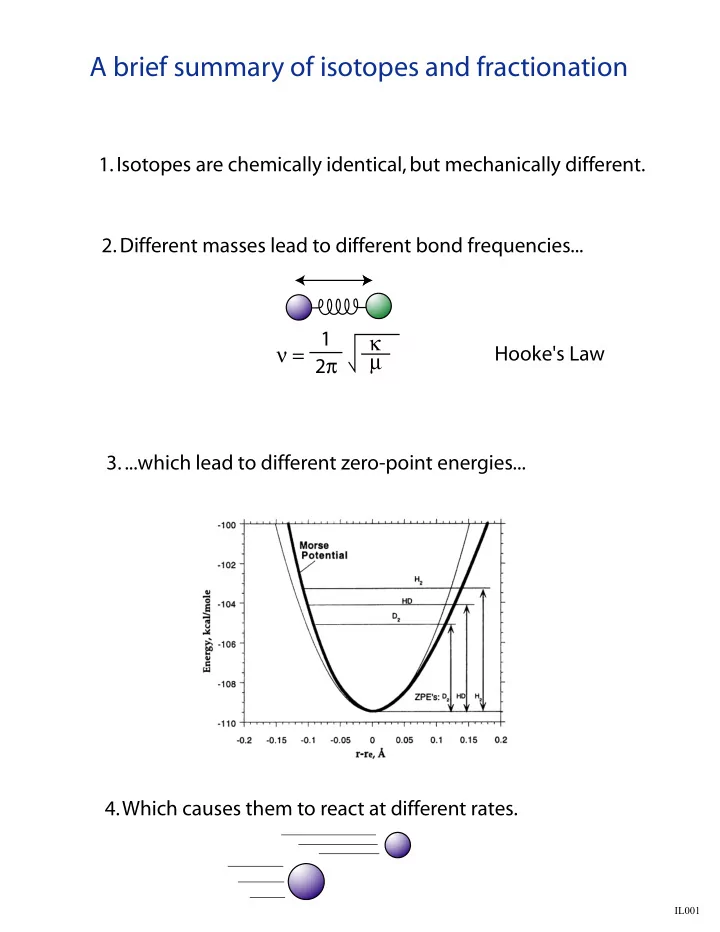
- 1. Isotopes are chemically identical, but mechanically different.
- 2. Different masses lead to different bond frequencies...
ν = 1 2π κ µ Hooke's Law
- 3. ...which lead to different zero-point energies...
- 4. Which causes them to react at different rates.
IL001