Takashi Kohno, PhD (tkkohno@ncc.go.jp)
Chief, Division of Genome Biology, Research Institute Chief, Division of Translational Genomics, Exploratory Oncology Research and Clinical Trial Center National Cancer Center , Tokyo, Japan 1
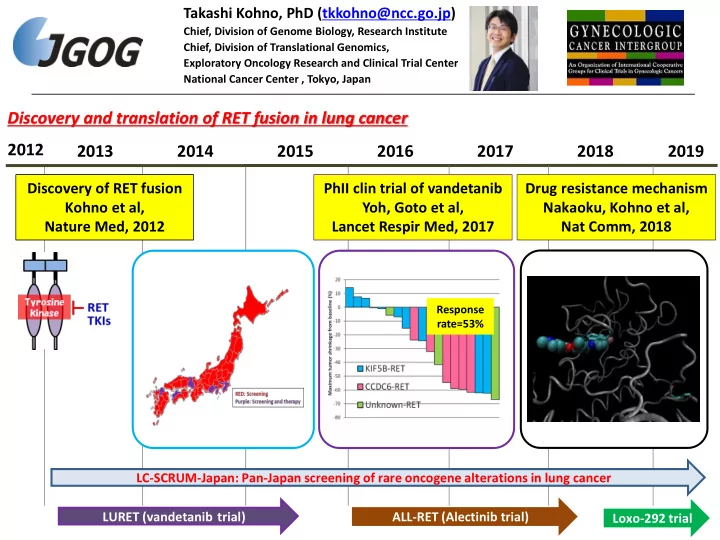
2012 2013 2015 2016 2017
LURET (vandetanib trial) ALL-RET (Alectinib trial)
Response rate=53%