L.I. Fatkhutdinova1, H. Bahrom2, A.A. Goncharenko3, O. Peltek1, A. Muslimov4, A. Manchev2,
- I. Shishkin1, R.E. Noskov2, A.S. Timin3, P. Ginzburg2 and M.V. Zyuzin1
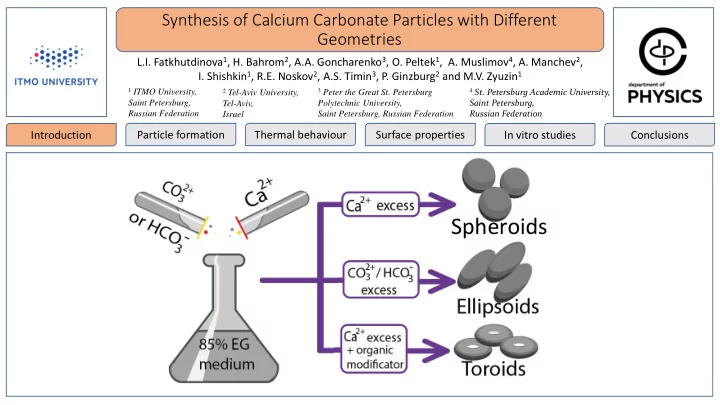
Figures, text etc (Column 1)
Spheroids
1 ITMO University,
Saint Petersburg, Russian Federation
2 Tel-Aviv University,
Tel-Aviv, Israel
3 Peter the Great St. Petersburg
Polytechnic University, Saint Petersburg, Russian Federation
4 St. Petersburg Academic University,
Saint Petersburg, Russian Federation
Synthesis of Calcium Carbonate Particles with Different Geometries
Introduction Particle formation Thermal behaviour Surface properties In vitro studies Conclusions