SLIDE 1
RACEMIZATION OF SECONDARY ALCOHOL OH OH Ru cat. R R Ru cat: - - PowerPoint PPT Presentation
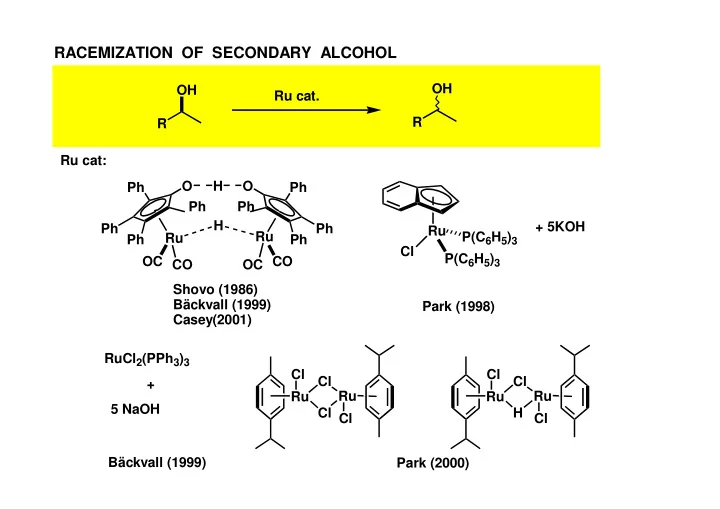
RACEMIZATION OF SECONDARY ALCOHOL OH OH Ru cat. R R Ru cat: - - PowerPoint PPT Presentation
RACEMIZATION OF SECONDARY ALCOHOL OH OH Ru cat. R R Ru cat: O H O Ph Ph Ph Ph H + 5KOH Ph Ph Ru Ru P(C 6 H 5 ) 3 Ru Ph Ph Cl P(C 6 H 5 ) 3 OC CO CO OC Shovo (1986) Bckvall (1999) Park (1998) Casey(2001) RuCl 2 (PPh
SLIDE 2
SLIDE 3
DEHYDROGENATIVE TRANSFORMATION OF ALCOHOLS
N Ru P H H H R C R' O Ar Ar N Ru P H H H R C R' O Ar Ar EPIMERIZATION OF meso-DIOLS ISOMERIZATION OF ALLYLIC ALCOHOLS LACTONIZATION OF α α α α,ω ω ω ω-DIOLS
SLIDE 4
ENANTIOMER DISCRIMINATION OF SECONDARY ALCOHOL
alcohol:Ru:chiral ligand:KOH = 100:1:1.5:1 R OH Cp*RuCl(cod) chiral ligand KOH 2-propanol, 30 °C R OH R = C6H5 or n-C4H9 kS/kR = 1.3 kS/kR = 1.8 R = n-C4H9 R = C6H5 * chiral ligand N P(C6H5)2 H
S
Ru N P(C6H5)2 amide complex
SLIDE 5
2 4 6 8 20 40 60 80 100 conversion, %
ENANTIOMER DISCRIMINATION OF sym-DIOL
time, h C6H5 C6H5 OH OH C6H5 C6H5 OH OH Cp*RuCl(cod) chiral ligand KOH 2-propanol, 30 °C hydrobenzoin:Ru:chiral ligand:KOH = 100:1:1.5:1 dl meso R,R-diol S,S-diol
R S
chiral ligand N P(C6H5)2 H
S
SLIDE 6
10 20 20 40 60 80 100 conversion, %
EPIMERIZATION OF meso-HYDROBENZOIN WITH ACHIRAL LIGAND
C6H5 C6H5 OH OH C6H5 C6H5 OH OH Cp*RuCl(cod) ligand KOH 2-propanol, 30 °C hydrobenzoin:Ru:ligand:KOH = 100:1:1.5:1 meso ligand H2N P(C6H5)2 reaction time, h
R S
* * dl
SLIDE 7
2 4 6 8 10 20 40 60 80 100 conversion and ee, %
EPIMERIZATION OF meso-HYDROBENZOIN WITH CHIRAL LIGAND
time, h C6H5 C6H5 OH OH C6H5 C6H5 OH OH Cp*RuCl(cod) chiral ligand KOH 2-propanol, 30 °C hydrobenzoin:Ru:chiral ligand:KOH = 100:1:1.5:1 chiral ligand N P(C6H5)2 H conv. ee
R S R R
meso dl
S
SLIDE 8
SYNTHESIS OF OPTICALLY ACTIVE DIOLs FROM meso-DIOLs
R OH R OH R OH R OH Cp*RuCl(cod) chiral ligand KOH 2-propanol, 30 °C, 4 h conv, % ee, % –(CH2)4– 49 18 99 98
- meso-diol:Ru:chiral ligand:KOH = 100:1:1.5:1
chiral ligand N P(C6H5)2 p-CH3-C6H4 conc. 16 91 0.1 M 0.05 M 0.1 M 0.1 M R C6H5 CH3 H
R R R S S
p-F-C6H4 22 85 0.1 M
SLIDE 9
A POSSIBLE MECHANISM FOR EPIMERIZATION
C6H5 C6H5 OH O C6H5 C6H5 OH OH C6H5 C6H5 OH OH C6H5 C6H5 O OH C6H5 C6H5 OH OH meso R,R S,S S R
R R R R S S S S
dl C6H5 C6H5 O OH
S
C6H5 C6H5 O OH KOH 2-propanol 30 °C, 1h S/C = 100
SLIDE 10