Project 1: Squam Contaminants
Photo: Lee Grenier
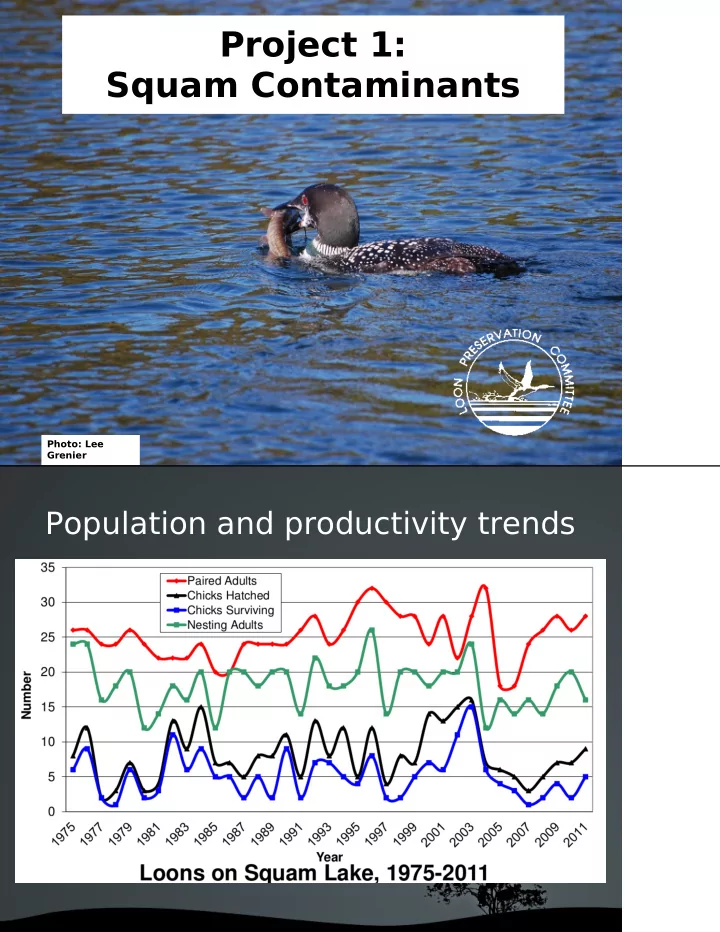
Project 1: Squam Contaminants Photo: Lee Grenier Population and - - PDF document
Project 1: Squam Contaminants Photo: Lee Grenier Population and productivity trends S q u a m L a k e L oon s O v e r T im e 16.0 Avg 1995-2004 14.0 Avg 2005-2007 12.0 Avg 2008-2012 10.0 8.0 6.0 4.0 2.0 0.0
Photo: Lee Grenier
0.0 2.0 4.0 6.0 8.0 10.0 12.0 14.0 16.0 Territorial Pairs Nesting Pairs C hicks Hatched C hicks S urviving
S q u a m L a k e L
s O v e r T im e
Avg 1995-2004 Avg 2005-2007 Avg 2008-2012
Figures from: http://www.missouriplants.com/Others/Myriophyllum_heterophyllum_plant.jpg https://c53a19f696-custmedia.vresp.com/library/1306354496/e2b7d3b0d8/uconn_ipane_myriospica_03c-reduced.JPG
Natives Bladderwort Coontail Fanwort Native Water Milfoil Pondweed Waterweed Non-Natives Brazillian elodea Euarasion Water-Milfoil Variable Water-Milfoil
CH 3600 / ESP 5090: Environmental Chemistry, Plymouth State University
12
CH 3600 / ESP 5090: Environmental Chemistry, Plymouth State University
13
CH 3600 / ESP 5090: Environmental Chemistry, Plymouth State University
14
CH 3600 / ESP 5090: Environmental Chemistry, Plymouth State University
15
CH 3600 / ESP 5090: Environmental Chemistry, Plymouth State University
16
CH 3600 / ESP 5090: Environmental Chemistry, Plymouth State University
17
Yes! No!
Measuring Volume Volumetric flasks Volumetric pipettes Auto-pipettes Burets Beakers Graduated cylinder Measuring Mass 4-digit Analytical balance RECORD ALL DIGITS Anything less than 4 digits Rounding last digit or 2
CH 3600 / ESP 5090: Environmental Chemistry, Plymouth State University
18