Natural Backgrounds
1
Natural Backgrounds 1 U 238 Decay Chain U 234 U 238 92 92 - - PowerPoint PPT Presentation
Natural Backgrounds 1 U 238 Decay Chain U 234 U 238 92 92 4.5e9 245,500 Years Years Uranium Pa 234 91 27 Days Protactinium Th Th 234 230 90 90 75,380 27 Days Years Thorium Actinium Ra 226 88
1
Protactinium Thorium Actinium Radium Francium Radon Astatine Polonium Bismuth Lead Thallium α α α β⁻ Uranium α β⁻ Mercury β⁻ α α α
Ra
226 1602 Years 88Th
234 27 Days 90Pa
234 27 Days 91Bi
214 20 Minutes 83Po
218 3.1 Minutes 84Pb
214 26.8 Minutes 82Tl
210 1.3 Minutes 81β⁻
At
218 1.5 85 SecondsPb
210 22.3 Years 82Po
214 0.1643 Seconds 84Th
230 75,380 90α α β⁻ β⁻
YearsRn
222 3.8 Days 86α
U
238 4.5e9 Years 92U
234 245,500 Years 92β⁻
Bi
210 83Tl
206 4.2 Minutes 81 5 DaysPb
206 Stable 82Po
210 138 Days 84α β⁻
Hg
206 8.1 Minutes 80α
Actinides Alkali Metals Alkaline Earth Metals Halogens Metalloids Noble Gases Poor Metals Transition MetalsThorium Actinium Radium Francium Radon Astatine Polonium Bismuth Lead Thallium α α α α α α
Actinides Alkali Metals Alkaline Earth Metals Halogens Metalloids Noble Gases Poor Metals Transition Metalsα β⁻ β⁻ β⁻
Rn
220
55 Seconds
86
Bi
212
61 Minutes
83
Po
212
3e-07
Seconds84
Pb
208
Stable82
Tl
208
3.1 Minutes
81
β⁻
Ac
228
Minutes
6.1
89
Ra
228
5.7 Years
88
Th
228
1.9 Years
90
α β⁻
Ra
224
3.6 Days
88
α
Po
216
0.14
Seconds84
Pb
212
10.6
Minutes82
Th
232
1.41e+10
Years
90
4
5
6
7 19
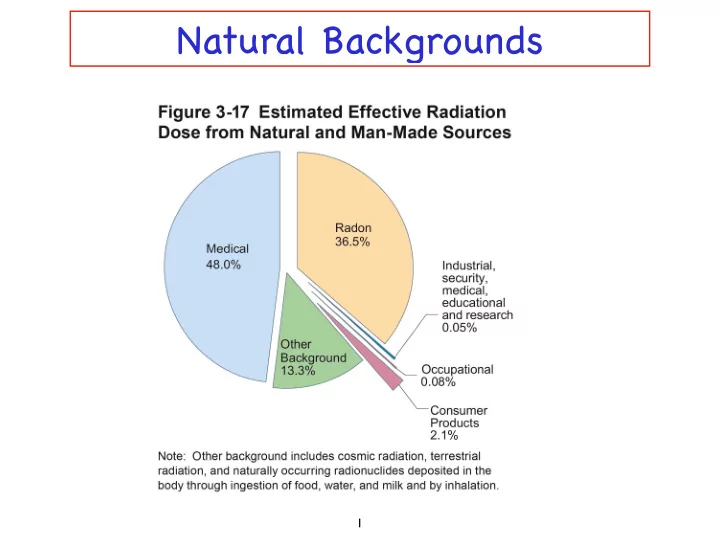
Heavy noble gas By product of uranium decay 200 mrem per year One in a hundred chance of inducing cancer over a person’ s lifetime Largest source of radiation exposure Main cause of lung cancer among nonsmokers
8
Average dose is 300 to 400 mrem (0.3 to 0.4 rem) Dose from living next to a nuclear power plant is 30,000 times less
9
10
11
12
Radiation can ionize (remove electrons from) atoms This breaks molecular bonds The DNA molecules that contain our genetic code are particular susceptible to radiation damage. DNA is a very fragile molecule. Its bonds can easily be broken.
If the gene on the DNA is mutated it can lead to the growth of caner cells
Harmful effects of radiation Cancer Radiation sickness / death
A large enough radiation exposure can cause large amount of cell death leading to sickness and, if enough, death
13
Below 100 rem no short term illness 100 to 200 rem short term illness Whole body doses 300 rem 50% chance of death More than 1000 rem survival unlikely
14
Whole body dose needed to for near certainty of inducing cancer
2500 rem
This is greater than the lethal dose! Even small doses have some probability of inducing cancer and it is likely cumulative over your lifetime Assume probability risk of cancer linearly proportional to dose Assume no threshold 25 rem exposure would mean a 1% risk of cancer If 100 people are exposed to 25 rem, we expect 1 to get cancer Even with no radiation exposure 20% of people will get cancer
15
16
Lifetime total natural exposure 1 in 100 (1.0%) Lifetime radon exposure 1 in 150 (0.7%) One medical x-ray 1 in 60,000 Lifetime self exposure 1 in 100,000 Living 50 years next to nuclear power plant 1 in 500,000 Non radiation causes 1 in 5 (20%)
Radiation from routine operation of a nuclear power plant is not a health issue
17
The nucleus at the center of each atom is made up of protons and neutrons Protons and neutrons have approximately equal masses and each is about 2000 times more massive than an electron. The number of protons in the nucleus is equal to the number of electrons in the electron cloud The atomic number, Z, is the number of protons The atomic weight, A, is the number of protons plus neutrons Nearly all of the mass of an atom is in the nucleus The nucleus is 100,000 times smaller than the atom If an atom were the size of the Rutgers football stadium the nucleus would be the size of a mosquito
18
In a chemical reaction electron binding energy is converted into kinetic energy Example the burning of ethane (a hydrocarbon) C C H H H H H H O O
+
O O
+
O H H 2 7 4 6 The daughter molecules are more tightly bound together than the parent molecules There is less chemical potential energy in the final state than in the initial. Chemical potential energy is converted to kinetic energy (heat) C2H6
+
O2 CO2
+
H2O
19
A nuclear reaction is similar
236U 92Kr
+
141Ba
+
3 neutrons The daughter nuclei are more tightly bound together than the parent nucleus There is less nuclear potential energy in the final state than in the initial. Nuclear potential energy is converted to kinetic energy (heat) Nuclear binding energy is 1 to 10 million time stronger than chemical binding energy
20
Because of the great difference in binding energies, for as given, mass, tremendous amount more energy is released in a nuclear reaction than in a chemical reaction Burning 1 gram of gasoline releases 42 kJ Fissioning 1 gram of Uranium releases 82 GJ In both cases we need a trigger. Ethane won’ t burn
t fission on its own In the case of a chemical reaction the trigger is a spark or heat In the case of a nuclear fission reaction the trigger is the nucleus being struck by an energetic neutron
21
Iron (Fe) is the most tightly bound nucleus Nuclei more massive than Fe can release energy by fissioning to lighter nuclei Nuclei less massive than Fe can release energy by fusing to form heavier nuclei
22
In a nuclear fission reaction a heavy parent nuclei fissions (or splits) into two or more lighter daughter nuclei. Several neutrons may be released in the process as well In general the heavier nuclei have a greater tendency to fission
235U Fission
23
If a neutron hits a 235U nucleus it will form a 236U nucleus that then fissions into lighter nuclei (fragments)
24
A chain reaction occurs if the fission process releases one or more neutrons that can then initiate addition fission reactions If a fission releases two neutrons after a few generations there will be many fissions. This all happens very rapidly
generation number of fissions 1 21 = 2 2 22 = 4 3 23 = 8 5 25 = 32 10 210 = 1000 20 220 = 106 40 240 = 1012 80 280 = 1024
25
If the chain reaction runs its course after 84 generations 10 kg of Uranium will have fissioned. 10 kg of Uranium corresponds to 300 kilotons of TNT Per weight, Uranium releases 30,000,000 times as much energy as TNT.
We have a bomb
In order for this to work two things are necessary. 1) Critical mass. There must be enough Uranium so that the neutrons interact before they escape. 2) Enrichment. There must a large enough fraction of 235U compared to 238U since 238U absorbs the neutrons. Critical mass: 15 kg Enrichment: 90%
26
The chain reaction can be controlled by using a material that absorbs neutrons By controlling the amount of absorber, we can arrange so that for every fission there in only
Then the reaction won’ t run away but will run at constant power. We have a nuclear reactor
27
Slow neutrons are much more effective in causing fission than fast neutrons Use a moderator that scatters neutrons but doesn’ t absorb them. An example is water After enough scattering the neutrons will be thermal. They will have energy corresponding to the ambient temperature.
28
29
Most reactors are moderated with light (normal) water Two types: pressurizer water (above) or boiling water. They require that the fuel be enriched to 3% to 4% 235U
30
Compacted fine powder of enriched Uranium oxide sintered into pellets and placed into long zircalloy tubes Fuel enriched to about 3% 235U About 100 tons of fuel per year for 1 GW reactor
31
Control rods usually contain boron since it is a very good neutron absorber
32
Naturally occurring uranium is only about 0.7% 235U Since the light water moderator also absorbs neutrons need to enrich to about 3% 235U. Heavy water moderator reactor uses unenriched fuel Enrichment needed for bomb is 20% minimum but efficient bomb requires 90% since don’ t want 238U to absorb the neutrons
33
Plutonium is the only other element that can undergo chain reaction fission It is more effective because it releases more neutrons per fission than uranium It is the element of choice for bombs Plutonium doesn’ t occur naturally but it is made in a nuclear reactor by absorption of a neutron by 238U
n + 238U --> 239U --> 239Np --> 239Pu
About 30% of the energy in a nuclear power reactor is from fission of plutonium, by the end of the fuel life cycle this becomes about 60%
34
Radiation Safety Waste disposal Cost Resource availability Weapons proliferation
35
36
Radiation Safety Waste disposal Cost Resource availability Weapons proliferation
37
Three Mile Island Chernobyl Fukushima
38
1979 Pennsylvania
carelessness
2 million people exposed to 1 mrem = 0.001 rem excess cancer deaths (2 x 106) x (0.001 rem) / (2500 rem) = 0.8
Reactors
39
2011 Japan
incompetence
far everything fine
venting gas and from water leaks
probably things would have been OK
Three Mile Island
deaths among exposed population of 5 million
40
1986 Ukraine
stupidity
would happen with cooling system disabled and control rods completely removed
less coolant
41
5 million people exposed to 100 mrem per year excess cancer deaths (5 x 106) x (5 rem) / (2500 rem) = 10,000 About 30% of core sent up into the atmosphere and spread over a wide area about one third of natural background
42
In assessing risks it is essential to take into account the number of people exposed Exposure in the Chernobyl closed zone (50 mile radius) is 3 rem per year 10 times normal background If you lived there for ten years you would have a 1% increased cancer risk. This is probably not worth worrying about. If 1,000,000 people lived there for ten years, there would be an additional 10,000 cancer deaths. This would be unacceptable. Bottom line. Exposure limits are set to protect the million. The risks for a single individual like you are very low. Same is of course true for non-radiation related risks
43
Reactors cannot blow up like nuclear bombs Chernobyl is the worst accident imaginable
large fraction of core sent into the atmosphere about 10,000 excess cancer death special to this type unstable flammable reactor
Modern reactors have advanced safety systems
gravity fed cooling works even if pumps shutdown
Accidents are probably unavoidable but we have seen the worst (Chernobyl) and it won’ t happen again
44
Radiation Safety Waste disposal Cost Resource availability Weapons proliferation
45
Fission Fragments
Long-lived Elements (Pu, Am, Np, Cu)
How to guarantee safe storage for 100,000 years or more
238U
46
radioactivity compared to uranium in the ground
1 million times greater when operating 100,000 times greater as soon as turned off 10,000 times greater after 2 months 100 times greater after 100 years < 1 times greater after 10,000 years
Doesn’ t include plutonium
47
Two options
total, and then secure the rest for a few hundred years.
48
Yucca Mountain was supposed to be the US solution
Deep stable geological repository in a mountain in Nevada remote, favorable climate, deep water table Limited by legislation to 70,000 tons. Could store much more After 20 years of intensive work, fell victim of politics Seems to be no hope for long term storage of this scale in US But need storage for 200,000 tons from current reactors
What do we do!!
This is a political not a technical or engineering problem
49
50
Fuel can be reprocessed to remove plutonium and other long-lived element Difficult but well established chemical process Several options after that
France is committed to this option US abandoned it because of nuclear proliferation issues
51
Radiation Safety Waste disposal Cost Resource availability Weapons proliferation
52
Immediately after Three Mile Island, nuclear power became factor of six more expensive Since then costs have come down dramatically
Nuclear power is now
Example: the AP1000 reactor shown in the Power Surge video
53
Radiation Safety Waste disposal Cost Resource availability Weapons proliferation
54
Estimated to be 3 million tons of uranium in the US 1 GW reactor uses about 200 tons per year US currently has 100 GW of operating capacity (3 million tons) (200 tons per GW/year) x (100 GW)
= 150 years
Uranium is not a long term solution Similar situation for rest of the world
55
n + 238U --> 239U --> 239Np --> 239Pu
Use reactor to breed (produce) plutonium from absorption of neutrons by 238U Effectively uses all of the uranium (including 238U) Extends lifetime of fuel resource by factor of 140
Problems:
56
Only four in the world 2 in France 1 in Kazakhstan 1 in Japan US abandoned this technology in the 1970’ s France plans on this for their energy future
57
Uranium enrichment Plutonium reprocessing
58
A nuclear reactor requires enriched uranium Any country that can enriched to reactor grade (3%) can also enrich to weapons grade (90%). Just run centrifuges longer 100 tons of uranium must be enriched for 1 year supply of fuel for a 1 GW reactor Only 1 ton of uranium need to be enriched for a nuclear bomb. If you have 1000 centrifuges for making reactor fuel, need only divert 10 of these for making a weapon
59
A nuclear reactor produces plutonium Any country with a nuclear reactor has plutonium A 1 GW reactor produces about 200 kg of 239Pu per year This is enough to produce about 100 plutonium bombs
60
A country with uranium enrichment or plutonium reprocessing capability can readily make nuclear weapons It is clear that these capabilities should not be widespread If uranium is to be enriched or plutonium to be reprocessed should be done in a neutral country under UN supervision. Has to be a reliable service that all countries of the world can trust This is a political, not scientific or technical, problem that the world has to solve if there is to be a future for nuclear energy
61
Radiation: not a health issue Safety: will be accidents but nothing like
what we’ve seen. Politically acceptable?
Waste disposal: issue is storing the 1% of waste that has a
long lifetime. This is a political not a technical
Cost: not now a problem. comparable to coal Resource availability: if to be long term solution need
breeder reactor technology. Expensive, dangerous, leads to proliferation problems
Weapons proliferation: major and real political problem
62
63
1H
2H
proton bound to one neutron. One part in 6000 of naturally
3H
proton bound to two neutrons. Unstable.
2H D 3H T
64
A different approach Instead of fissioning a heavy nucleus into lighter
per weight than Uranium fission
(hydrogen or heavy hydrogen)
65
In order to get hydrogen to fuse the nuclei must be at high temperature to overcome the barrier due to electrical repulsion. Kind of like pushing something uphill to get it into a valley
need high temperature to push uphill energy released when fall into valley
Temperature needed is about 100 million degrees. Ten times greater than those in the center of the Sun Fusion is the process that fuels the Sun
66
The Sun fuses two protons together. Fusing heavy hydrogen requires somewhat less temperature and is much easier approach for a fusion reactor D-D Reaction D + D 3He + n D-T Reaction D + T 4He + n The D-T reaction is the easiest to achieve and will be the first to be demonstrated in a test fusion reaction Tritium does not occur naturally and is somewhat dangerous. Why? A power plant reactor will have to be based on the D-D reaction
67
s oceans is enough to supply all of the world’ s energy needs for a billion years
negligible compared to the radioactivity from a fusion reactor
This is all fantastic. Now, how do we get it to work
68
Hydrogen in the from of a plasma (gas of charged particles) Plasma confined by a toroidal (doughnut) magnetic field Plasma can also be heated by magnetic field magnetic field Can’ t let plasma touch anything. It will loose all of its heat and become cold
Problem is instabilities.
After very short time loose control of the plasma Huge engineering problem Might be insoluble
69
70
International Thermonuclear Experimental Reactor
Designed to produce 10 times more power than it consumes But only briefly. 500 MW but only for up to 10 minutes Engineering demonstration. Not a power reactor D-T reaction Cost: about $20 billion
71
Joke is that fusion reactors are a great idea but fusion power is always about fifty years away. I would say now it is more like 100 years away. Maybe by 2100
Timeline Date Event 2006-11-21 Seven participants formally agreed to fund the creation of a nuclear fusion reactor.[9] 2008 Site preparation start, ITER itinerary start.[20] 2009 Site preparation completion.[20] 2010 Tokamak complex excavation start.[21] 2011 Tokamak complex construction start.[21] 2015 Predicted: Tokamak assembly start.[12] 2018 Predicted: Tokamak assembly completion, start torus pumpdown.[12] November 2019 Predicted: Achievement of first plasma.[22] 2026 Predicted: Start of deuterium-tritium operation.[22] 2038 Predicted: End of project. ITER Timeline