MONALEESA-2 Interim Analysis: Ribociclib Plus Letrozole
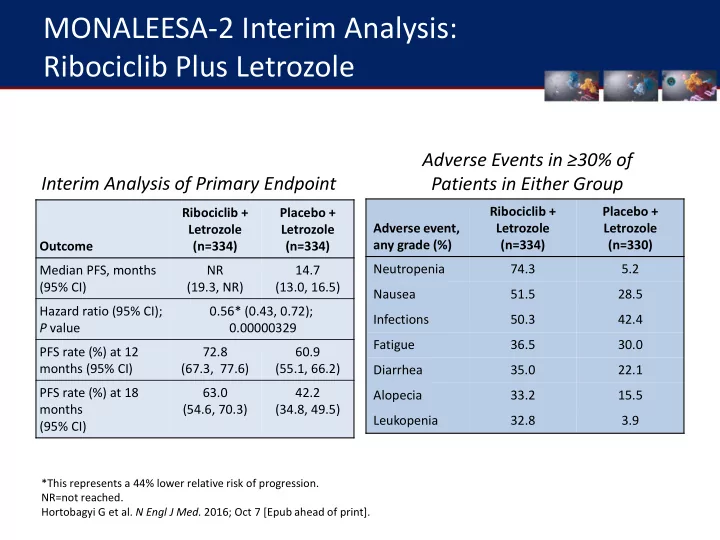
Outcome Ribociclib + Letrozole (n=334) Placebo + Letrozole (n=334) Median PFS, months (95% CI) NR (19.3, NR) 14.7 (13.0, 16.5) Hazard ratio (95% CI); P value 0.56* (0.43, 0.72); 0.00000329 PFS rate (%) at 12 months (95% CI) 72.8 (67.3, 77.6) 60.9 (55.1, 66.2) PFS rate (%) at 18 months (95% CI) 63.0 (54.6, 70.3) 42.2 (34.8, 49.5)
*This represents a 44% lower relative risk of progression. NR=not reached. Hortobagyi G et al. N Engl J Med. 2016; Oct 7 [Epub ahead of print].
Interim Analysis of Primary Endpoint Adverse Events in ≥30% of Patients in Either Group
Adverse event, any grade (%) Ribociclib + Letrozole (n=334) Placebo + Letrozole (n=330) Neutropenia 74.3 5.2 Nausea 51.5 28.5 Infections 50.3 42.4 Fatigue 36.5 30.0 Diarrhea 35.0 22.1 Alopecia 33.2 15.5 Leukopenia 32.8 3.9