SLIDE 4 November 26, 2018 4
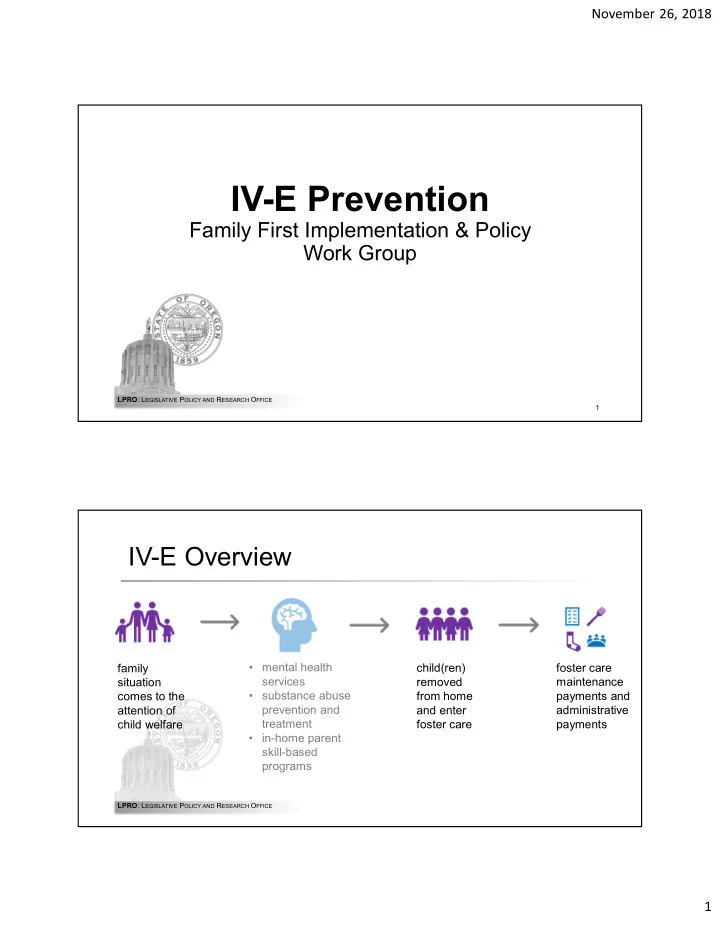
Prevention Program Requirements
LPRO: LEGISLATIVE POLICY AND RESEARCH OFFICE
there is no case data suggesting a risk of harm that was probably caused by the treatment and that was sever or frequent practice has a book, manual, or
writings that specify the components of the practice protocol and describe how to administer the practice there is no empirical basis suggesting that, compared to its likely benefits, the practice constitutes a risk of harm to those receiving it if multiple outcome studies have been conducted, the
evidence supports the benefits of the practice
measures are reliable and valid, and are administered consistently and accurately across all those receiving the practice
Evidence Standard
LPRO: LEGISLATIVE POLICY AND RESEARCH OFFICE
Promising: At least one study that used some form of control group (e.g., wait list study, placebo group) to determine effect. Supported: At least one study that used a random control or quasi- experimental trial to determine effect; sustained effect of at least 6 months. Well-supported: At least two studies that used a random control or quasi- experimental trial to determine outcomes; sustained effect of at least 1 year. Services and programs that meet the requirements for being a promising, supported, or well-supported practice shall be IV-E eligible.