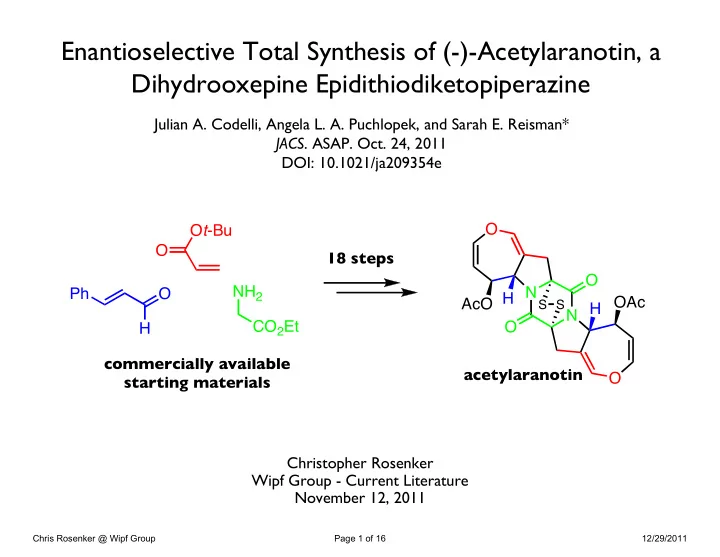
Julian A. Codelli, Angela L. A. Puchlopek, and Sarah E. Reisman*
- JACS. ASAP. Oct. 24, 2011
DOI: 10.1021/ja209354e Christopher Rosenker Wipf Group - Current Literature November 12, 2011
Enantioselective Total Synthesis of (-)-Acetylaranotin, a Dihydrooxepine Epidithiodiketopiperazine
O N N O AcO H O O H OAc
S S
18 steps Ot-Bu O O H Ph NH2 CO2Et commercially available starting materials acetylaranotin
Chris Rosenker @ Wipf Group Page 1 of 16 12/29/2011