SLIDE 1
1
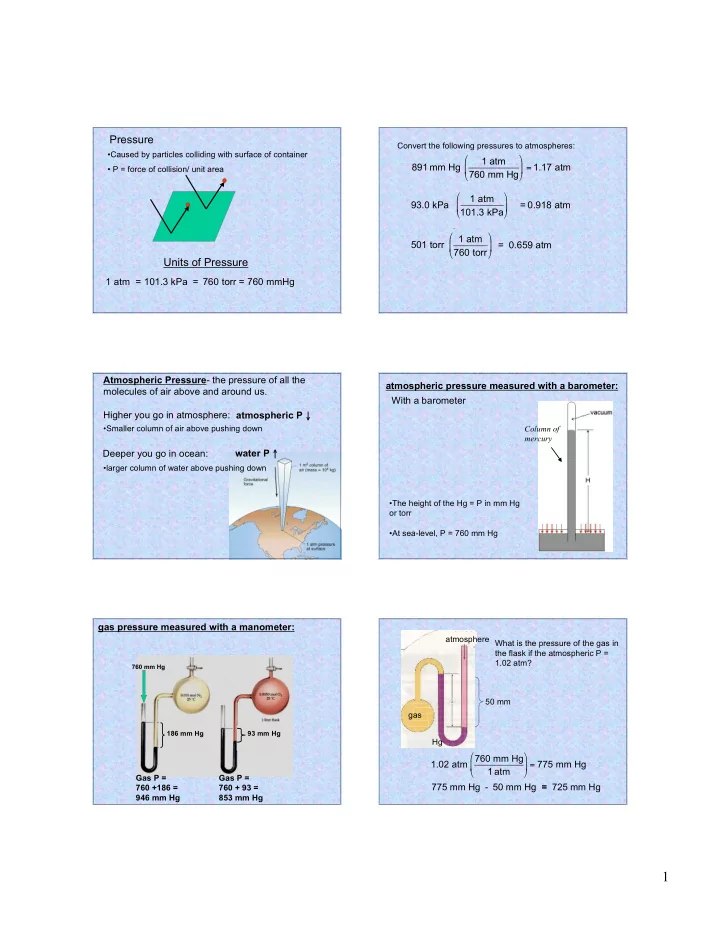
Pressure
- Caused by particles colliding with surface of container
- P = force of collision/ unit area
Units of Pressure
1 atm = 760 torr = 760 mmHg 101.3 kPa =
Convert the following pressures to atmospheres:
891 mm Hg 93.0 kPa 501 torr mm Hg 1 atm 760 mm Hg
- = 1.17 atm
93.0 kPa 1 atm 101.3 kPa
- = 0.918 atm
- 501 torr
1 atm 760 torr
- = 0.659 atm
Atmospheric Pressure- the pressure of all the molecules of air above and around us. Higher you go in atmosphere: atmospheric P ↓ Deeper you go in ocean: water P ↑
- Smaller column of air above pushing down
- larger column of water above pushing down
Column of mercury
atmospheric pressure measured with a barometer:
- The height of the Hg = P in mm Hg
- r torr
- At sea-level, P = 760 mm Hg
With a barometer gas pressure measured with a manometer:
760 mm Hg 186 mm Hg 93 mm Hg
Gas P = 760 +186 = 946 mm Hg Gas P = 760 + 93 = 853 mm Hg 50 mm What is the pressure of the gas in the flask if the atmospheric P = 1.02 atm? Hg atmosphere gas
1.02 atm atm 760 mm Hg 1 atm
- = 775 mm Hg