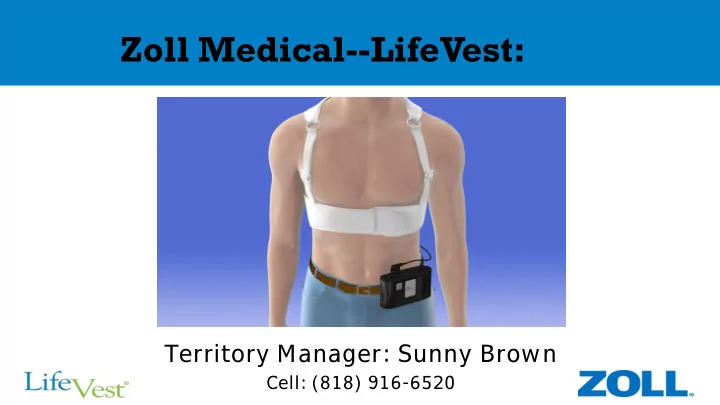
Zoll Medical--LifeVest:
Territory Manager: Sunny Brown
Cell: (818) 916-6520
Zoll Medical--LifeVest: Territory Manager: Sunny Brown Cell: (818) - - PowerPoint PPT Presentation
Zoll Medical--LifeVest: Territory Manager: Sunny Brown Cell: (818) 916-6520 Objectives Why the LifeVest device exist Review indications for Wearable Cardioverter Defibrillator (WCD) use Give a brief description of the LifeVest
Cell: (818) 916-6520
http://www.youtube.com/watch?v=gFLfNao-rR8
(1) Nichol et al. Circ, April 21, 2008. (2) Peberdy, et al, RESUSCITATION 58 (2003) 297-308 (3) Valenzuela et al., NEJM. Oct 26, 1206-9, 2000.
(1)AHA Heart Disease and Stroke Statistics-2015 Update
(1)Myerburg RJ, Juntilla MJ. Sudden cardiac death caused by coronary heart disease. Circulation 2012;125:1043-52 Goldberger JJ, Buxton AE, Cain M, et al. Risk stratification for arrhythmic sudden cardiac death: identifying the roadblocks. Circulation. 2011;123:2423-30. (2) Fishman GI, Chugh SS, DiMarco JP, et al. Sudden cardiac death prediction and prevention: report from a National Heart, Lung, and Blood Institute and HRS Workshop. Circulation. 2010;122:2335-48. Myerburg RJ. Sudden cardiac death: exploring the limits of our knowledge. J Cardiovascular Electrophysiol. 2001;12:369-81.
the home, survival rate is 6%
(1)
(1) Merchant RM, Yang L, Becker LB et al. Incidence of treated cardiac arrest in hospitalized patients in the United States. Crit Care Med. 2011;39:2401-6. (2) Daya MR, Schmicker R, MaySH, et al. Current burden of cardiac arrest in the United States: report from the Resuscitation Outcomes Consortium. Paper commissioned by the Committee on the Treatment of Cardiac Arrest: Current Status and Future Directions. 2015.
Who doesn't receive SCD protection?
Gel capsules Defibrillation electrode ECG electrode
SS FB SS FB SS FB SS FB
shock recovery
WCD = LifeVest
WCD = LifeVest
The LifeVest is covered by most health plans in the United States, including commercia, state and federal
specific health plan coverage policy should be reviewed to determine coverage for prescribing a LifeVest
period)
until no longer at risk
until no longer at risk
drug-eluting stents
deployment of a drug eluting stent
converted to NSR by a LifeVest shock
physiological risk factors to protect to provide protection from SCA
6
Kooshkabadi Case Study
unable to terminate VF. He required simultaneous 300 joule shocks from two separate AEDs during the EP study.
VF with a single 150 joule shock. He was home for Christmas.