CLEAN COMBUSTION RESEARCH CENTER
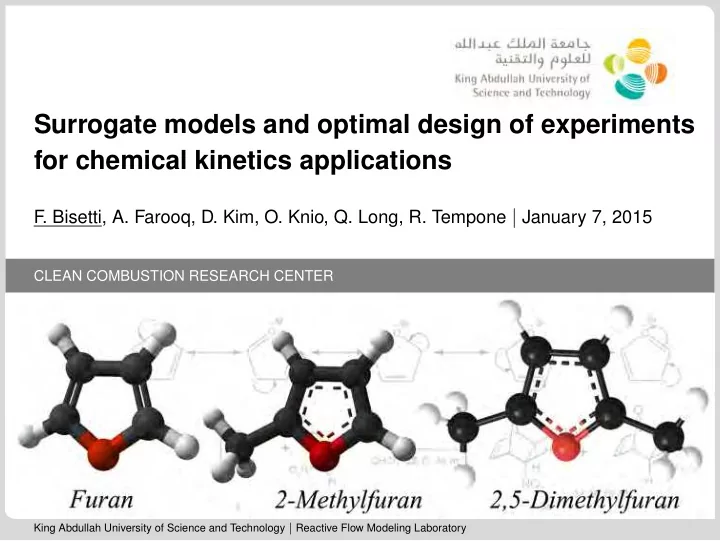
Surrogate models and optimal design of experiments for chemical kinetics applications
F . Bisetti, A. Farooq, D. Kim, O. Knio, Q. Long, R. Tempone | January 7, 2015
King Abdullah University of Science and Technology | Reactive Flow Modeling Laboratory