Specific Heats of Ideal Gases
Assume that a pure, ideal gas is made of tiny particles that bounce into each other and the walls of their cubic container of side ℓ. Show the average pressure P exerted by this gas is P = 1 3 N V mv 2
total
Use the ideal gas law (PV = NkBT = nRT)and the conservation
- f
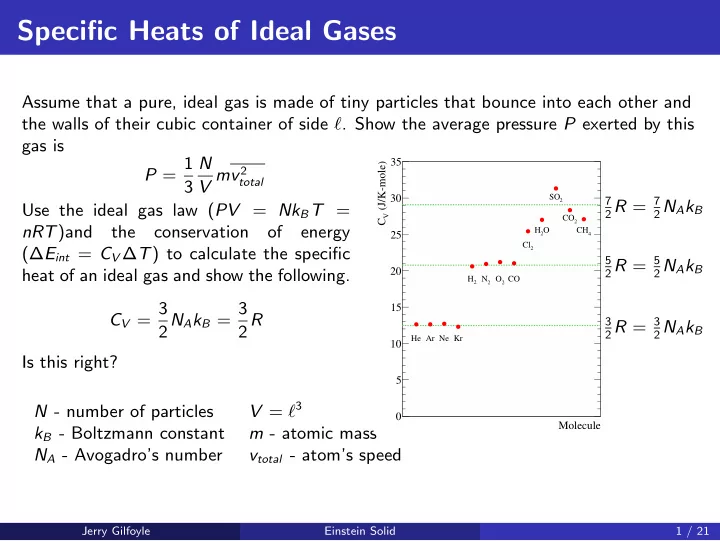
energy (∆Eint = CV ∆T) to calculate the specific heat of an ideal gas and show the following. CV = 3 2NAkB = 3 2R Is this right? N - number of particles V = ℓ3 kB - Boltzmann constant m - atomic mass NA - Avogadro’s number vtotal - atom’s speed
Molecule (J/K-mole)
V
C 5 10 15 20 25 30 35
He Ar Ne Kr
2
H
2
N
2
O CO
2
Cl O
2
H
2
SO
2
CO
4
CH
3 2R = 3 2NAkB 5 2R = 5 2NAkB 7 2R = 7 2NAkB Jerry Gilfoyle Einstein Solid 1 / 21