1
Physics 2D Lecture Slides Lecture 12: Jan 26th 2005
Vivek Sharma UCSD Physics
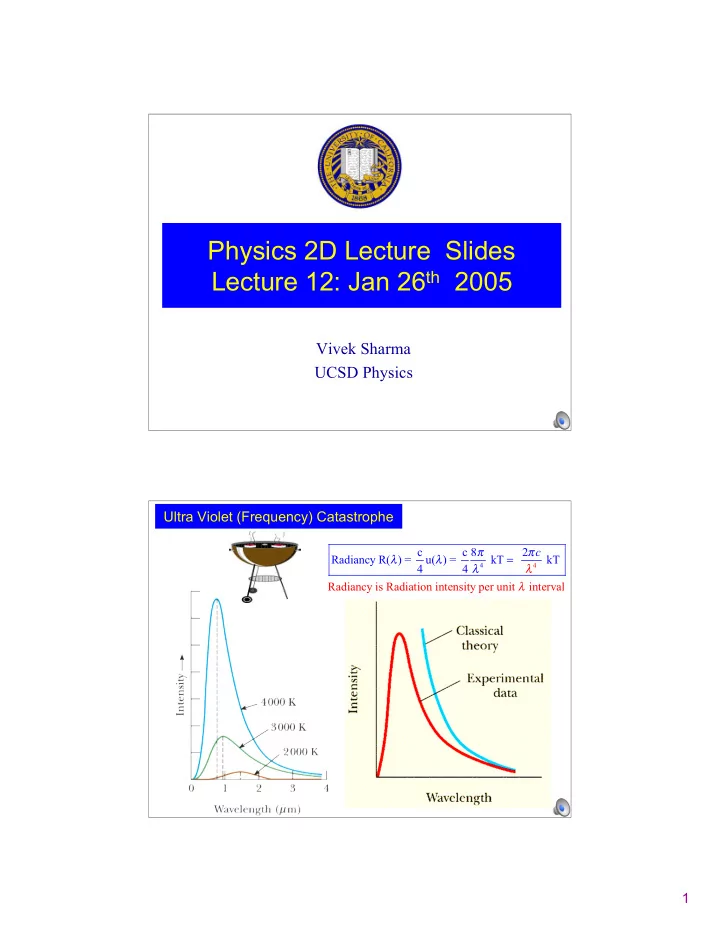
Ultra Violet (Frequency) Catastrophe
4 4
Radianc c c 8 2 Radiancy y is Radiatio R( ) = u( ) = kT n intensity per u k nit interval T 4 4 c
- =
Physics 2D Lecture Slides Lecture 12: Jan 26 th 2005 Vivek Sharma - - PDF document
Physics 2D Lecture Slides Lecture 12: Jan 26 th 2005 Vivek Sharma UCSD Physics Ultra Violet (Frequency) Catastrophe c c 8 2 c = Radiancy R( ) = u( ) = kT k T 4 4 4 4 Radianc y is Radiatio n
1
Ultra Violet (Frequency) Catastrophe
4 4
Radianc c c 8 2 Radiancy y is Radiatio R( ) = u( ) = kT n intensity per u k nit interval T 4 4 c
2
i Light of intensity I, wavelength λ and frequency ν incident on a photo-cathode Measure characteristics of current in the circuit as a fn of I, f, λ
– From current i seen in ammeter
– By applying retarding potential on electron moving towards Collector plate
3 Observation: Photo-Current Vs Frequency of Incident Light
I3 = 3I1 I2 = 2I1 I1= intensity f
Stopping voltage VS is a measure of the Max kinetic energy of the electron
Stopping Voltage Vs For Different Photocathode Surfaces eVS = KMAX = max KE
4
Shining Light With Constant Intensity But different frequencies Larger the frequency of light, larger is the stopping voltage (and thus the kinetic energy of the “photoelectrons” ) f1 > f2 >f3
f1 f2 f3
Current i in circuit
I
– f0 is characteristic of that metal
5
– E field and electrical force seen by the “charged subatomic oscillators” Larger
more Kinetic Energy KE !! The intensity of light shining rules !
cause photoelectric effect
Spherical wavefront incident on cathode, thould be a noticeable time lag ΔT between time is incident & the time a photo-electron is ejected : Energy absorption time
– How much time ? Lets calculate it classically.
size of atom in cathode metal
minimum amount of radiation before its stripped off
– Binding energy = 2.3 eV= “Work Function” – Electron confined in Na atom, size ≅ 0.1nm ..how long before ejection ?
– Average Power Delivered PAV = I . A, A= πr2 ≅ 3.1 x 10-20 m2 – If all energy absorbed then ΔE = PAV . ΔT ⇒ ΔT = ΔE / PAV – Classical Physics predicts Measurable delay even by the primitive clocks of 1900 – But in experiment, the effect was observed to be instantaneous !!
– Classical Physics fails in explaining all results
19 2 20 2
6
Planck noted the UltraViolet Catastrophe at high frequency “Cooked” calculation with new “ideas” so as bring: R(λ) 0 as λ 0 f ∞ Back to Blackbody Radiation Discrepancy
radiation & “atomic” oscillators present on walls of cavity
continuous and arbitarary…it is discrete …in packets of same amount
h = constant he invented, a very small number he made up
7 Planck’s “Charged Oscillators” in a Black Body Cavity Planck did not know about electrons, Nucleus etc: They were not known
possible Energy of wave is distributed over a spectrum of states: P(E) = e(-E/kT)
hf P(E) E e(-E/kT) By this statistics, large energy, high f modes of EM disfavored
8
2 x 2 4 3
8 ( ) 4 Odd looking form hc When large small kT 1 1 1 1 ( ....] Recall e 1 1 1 .... 2! 2 = 3!
hc kT hc kT
hc e hc hc e kT kT h x c c x R x
+ + + +
8 plugging this in R( ) eq: ) ( 4 c R kT hc kT
With BBQ data
4
hc kT h hc hc kT kT c k c kT T h
9
10
Amongst his lesser known talents was his ability to communicate. here he is greeting old friend: Conrad Habicht What are you up to? you frozen whale, you smoked, dried, canned piece of soul Clearly , like the electron, the phrase “ Whaddup Dog !” had not been discovered by then !
Light as bullets of “photons” Energy concentrated in photons Energy exchanged instantly Energy of EM Wave E= hf What Maxwell Saw of EM Waves What Einstein Saw of EM Waves
11
PHOTON
energy which depends on frequency f
– Transfer all energy to electron – Energy gets used up to pay for Work Function Φ (Binding Energy)
function of electron in atom
I3 = 3I1 I2 = 2I1 I1= intensity Light shining on metal cathode is made of photons Energy E, depends on frequency f , E = hf = h (c/λ) This QUANTUM of energy is used to knock off electron
electro s n
12
Light shining on metal cathode is made of photons Quantum of Energy E = hf = KE + ϕ ⇒ KE = hf - ϕ Shining Light With Constant Intensity f1 > f2 >f3
f1 f2 f3
13
No matter where you travel in the galaxy and beyond… ..no matter what experiment You do h : Planck’s constant is same NOBEL PRIZE FOR PLANCK
14
2 2
Light of Intensity I = 1.0 W/cm inc A Photoelectric Effect on An Iron Surfa ssume Fe reflects 96% of ligh ce: further on ident on ly 3% of 1.0cm surfa incident li ce of ght i i F t e s V µ
2
(a) Intensity available for Ph. El eff I =3
barely above thres ect (b) how m hold frequency for Ph any photo-electrons e . El effec mitted per t # s % 4% (1.0 W/c econd ? m )
9 34 2 9
Power = h f hc (250 10 )(1.2 10 / ) = (6.6 10 )(3.0 1 p 3% 4 / ) hoto % (1.0 W/c electro m n ) s m J s J s m s µ
10
9 15 1
9
= (c) Current in Ammeter : i = (1.6 10 )(1.5 10 ) (d) Work Function = ( )( ) 2.4 10 h 4.14 1 1.5 10 f 1.1 10 = 4.5 eV C A s eV s
– Energy to Dissociate an AgBr molecule = 0.6eV
– chlorophyll absorbs best at λ ≅ 650-700 nm
15
– ⇒ p = h/λ
– Momentum of the photon (light) is inversely proportional to λ
Useful for probing structure of sub-atomic Particles (and your teeth)
16
when electron passes near a positively charged target nucleus contained in target material, its deflected from its path because of its electrical attraction , experiences acceleration. Rules of E&M say that any charged particle will emit radiation when accelerated. This EM radiation “appears” as photons. Since photo carries energy and momentum, the electron must lose same amount. If all of electron’s energy is lost in just one single collision then
max min min
= hf
hc hc e V e V
=
continuously
the nuclear structure of target material and varies between materials
Molybdenum (Mo)
grating is too wide, need smaller slits
grating (Bragg)
17
– Interference – Diffraction
– Good Resolution λ<< Δ
properties (acts like photons)
– Showed that classical theory failed to explain the scattering effect of
– Watch the scattering of light off electron : measure time + wavelength of scattered X-ray
18
a surface with loosely bound electrons interaction of E field of EM wave with electron: F = eE
f = fincident
spherical waves with fradiated= fincident
– At all scattering angles, Δf & Δλ must be zero
electron gets a “tan” : soaks in radiation
( ' ) (1 cos ) Scattered ' larger than incident
19
How does one explain this startling anisotropy?
'
20
2 e e e
E+m ' p = p'cos +p cos p'sin -p sin Use these to e Energy Conservation: Momentum Conserv liminate electron deflection angle (n
: )
e
c E E
+ =
e e e 2 2 2 2 4 2 e 2 2 e e 2
p 2 'cos p cos 'cos p sin 'sin Square and add Eliminate p & using E & E ( ') '
e e e e
p c m c E E m p p p E p pp p c
=
+ = = +
21
2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2 2
( ') ' 2 ' 2( ') ( ' ) ( 2 'cos ( ) E For light p= c ' ( ') 'cos E-E' 1 )(1 co ' ' ' 2 co (1 cos ) EE' s s )
e e e e
E E m c EE E E m c E E EE E mc p pp p E E E E E mc h E E c c c c m m c c
+
= +
+
2 e e e
E+m ' p = p'cos +p cos p'sin -p sin Use these to e Energy Conservation: Momentum Conserv liminate electron deflection angle (n
: )
e
c E E
+ =
22
Plot scattered photon data, calculate slope and measure “h”
( ' ) ( )(1 cos )
e
h m c
Energy Quantization is a UNIVERSAL characteristic
' max
Two Identical waves travel along +x and interefere to give a resulting wave y ( , ). The resulting wave form depends on relative phase differen ( , ) sin(
ce between 2 waves. Shown f
i i i i
y x t y t x x k t
+ 2 = 0 r 3
etal held in Ereserve
23
The “High Energy Accelerator” of 1900s: produced energetic light : X Ray , gave new optic to subatomic phenomena Xray e
24 Bragg Scattering: Probing Atoms With X-Rays Constructive Interference when net phase difference is 0, 2π etc This implied path difference traveled by two waves must be integral multiple of wavelength : nλ=2dsinϑ
25 Proteins inside Rhinovirus reconstructed by x-ray diffraction