SLIDE 17 17
EE311/ Cu Interconnect
33 tanford University araswat
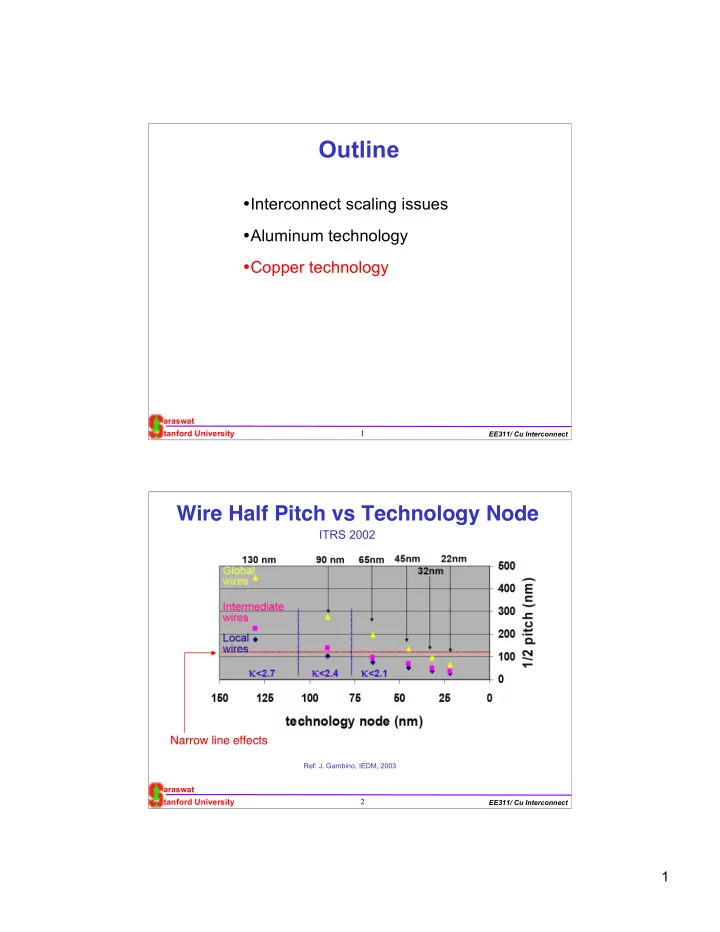
Semi-global & Local Interconnects
Kapur, McVittie & Saraswat, IEEE Trans. Electron Dev. April 2002
Temp.=100 0C Technology node (µm)
Al P=0 P=0.5 P=1 Cu, P=0.5
0.18 0.15 0.12 0.1 0.07 0.05 0.035
PVD C-PVD A L D : 1 n m A L D : 3 n m A L D : 1 n m No Barrier
Local
Year
Effective resistivity (µ ohm-cm) Local Temp.=100 0C
- With ALD least resistivity rise
- Al resistivity rises slower than Cu. Cross over with Cu resistivity possible
– no 4 sided barrier, needs only thin TiN to improve reliability and as anti reflection coating – smaller λmfp => smaller k – But has reliability problem Al
Cu
Technology node (µm)
Al
P=0 P=0.5 P=1
Cu, P=0.5
0.18 0.15 0.12 0.1 0.07 0.05 0.035
PVD C-PVD I
V D ALD: 10nm ALD: 3nm ALD: 1nm N
a r r i e r
Semiglobal Temp.=100 0C
EE311/ Cu Interconnect
34 tanford University araswat
- Higher temperature ⇒ lower mobility ⇒ higher resistivity
- Realistic Values at 35 nm node: P=0.5, temp=100 0C
- local ~ 5 µΩ-cm
- semi-global ~ 4.2 µΩ-cm
- global ~ 3.2 µΩ-cm
Cu Resistivity: Effect of Chip Temperature
2000 2004 2008 2012 Year 0.18 0.12 0.07 0.05 Technology Node (µm) 0.035 3.6 3.2 2.4 1.6 Effective resistivity (microohm-cm) 2 2.8
T=100 0C T=27 0C
Global
Kapur, McVittie & Saraswat IEEE Trans. Electron Dev. April 2002